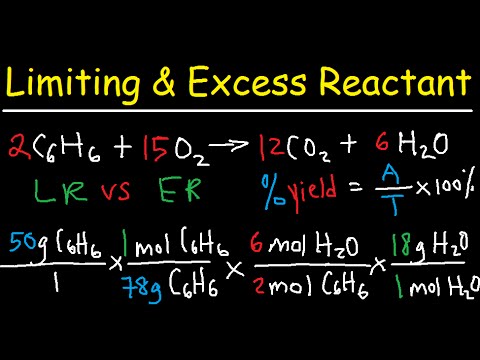IB Chemistry Topic 1 Stoichiometric relationships Topic 1.3 Reacting masses and volumes SL
Summary
TLDRThis comprehensive chemistry tutorial covers essential topics like mole calculations, percent yield, limiting reactants, and gas laws. It explains how to calculate theoretical yields and identify limiting reagents using stoichiometry, as well as explore gas behavior through laws such as Boyle's, Charles', and the Ideal Gas Law. The script also delves into practical laboratory techniques, including concentration calculations, titrations, and proper dilution practices. Key concepts are reinforced with real-world problems, ensuring a solid understanding of both theoretical and practical chemistry principles.
Takeaways
- 😀 Percent yield and percent error can be calculated by comparing the theoretical amount with the actual amount obtained in a chemical reaction.
- 😀 The limiting reactant determines how much product can be formed in a reaction; it’s the reactant that will run out first.
- 😀 In stoichiometric calculations, start by converting given masses into moles, then use the balanced equation to find the limiting reactant.
- 😀 Gas volume and moles are related at standard temperature and pressure (STP), with 1 mole of gas occupying 22.7 liters.
- 😀 Ideal gas laws assume no intermolecular forces, that particles are in random motion, and that collisions are perfectly elastic.
- 😀 Gas laws like Boyle’s, Charles’s, and Gay-Lussac’s describe the relationship between pressure, volume, and temperature of gases.
- 😀 The Ideal Gas Law (PV = nRT) allows you to calculate various properties of gases, but requires unit conversions (e.g., pressure in pascals, volume in cubic meters, temperature in Kelvin).
- 😀 Dalton's Law of Partial Pressures states that the total pressure of a gas mixture is the sum of the partial pressures of its individual gases.
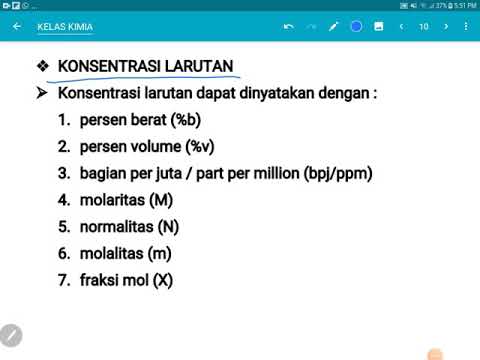
- 😀 Concentration calculations can be done using the formula C = moles/volume, with molarity (moles per liter) being a common unit.
- 😀 Titrations are used to determine the concentration of an unknown solution by neutralizing it with a solution of known concentration (titrant).
- 😀 Dilution calculations involve using the equation C1V1 = C2V2 to prepare solutions of lower concentration from more concentrated ones.
Q & A
What is percent yield and how do you calculate it?
-Percent yield is a measure of the efficiency of a reaction. It is calculated by dividing the actual yield (the amount of product obtained from a reaction) by the theoretical yield (the amount of product predicted by stoichiometry), and multiplying by 100. For example, if the theoretical yield is 51 g and the actual yield is 45 g, the percent yield is 88%.
Why is it important to identify the limiting reactant in a chemical reaction?
-The limiting reactant is important because it determines how much product can be formed. The reaction stops when the limiting reactant is used up, and no more product can be made, regardless of how much of the other reactants are available.
How do you determine the limiting reactant in a reaction?
-To determine the limiting reactant, you need to calculate the number of moles of each reactant. Then, using the stoichiometric ratio from the balanced equation, you can determine which reactant will run out first, thus limiting the amount of product that can be formed.
What is the relationship between volume and temperature for gases, according to Charles' law?
-According to Charles' law, the volume of a gas is directly proportional to its temperature, provided the pressure is constant. As the temperature increases, the volume of the gas also increases, and vice versa.
What are the assumptions made about ideal gases?
-Ideal gases are assumed to have particles in random motion, with negligible volume compared to the space between them. There are no forces of attraction between particles, and collisions between particles and the container walls are perfectly elastic. The average kinetic energy of the particles is directly proportional to the temperature in Kelvin.
How do you convert between different units when using the ideal gas law?
-When using the ideal gas law (PV = nRT), it's important to use the correct units: pressure in pascals (Pa), volume in cubic meters (m³), temperature in Kelvin (K), and the gas constant (R) in appropriate units. Unit conversions may include converting pressure from atmospheres to pascals and volume from decimeters cubed to meters cubed.
What is Dalton's law of partial pressures?
-Dalton's law states that the total pressure of a mixture of gases is the sum of the partial pressures of the individual gases. Each gas exerts a pressure based on its proportion in the mixture, and these individual pressures can be added together to find the total pressure.
How do you calculate the concentration of a solution?
-Concentration is calculated by dividing the number of moles of solute by the volume of the solution. It is typically expressed in moles per liter (mol/L), also known as molarity. The formula is C = n/V, where C is concentration, n is the number of moles, and V is the volume of the solution.
What is the formula for diluting a solution, and how is it used?
-The formula for diluting a solution is C1V1 = C2V2, where C1 and V1 are the concentration and volume of the stock solution, and C2 and V2 are the desired concentration and volume of the diluted solution. This formula allows you to calculate how much of the stock solution is needed to prepare a diluted solution.
What is the procedure for performing a titration, and how is it used to determine concentrations?
-In a titration, a solution of known concentration (titrant) is gradually added to a solution of unknown concentration (analyte) until the reaction reaches its endpoint, often indicated by a color change. By using the volume of titrant required and the balanced equation, you can calculate the concentration of the analyte.
Outlines

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифMindmap

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифKeywords

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифHighlights

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифTranscripts

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифПосмотреть больше похожих видео
5.0 / 5 (0 votes)