16.1 Thermochemistry
Summary
TLDRThis educational video script covers thermochemistry, focusing on energy transfer in chemical reactions. It explains the difference between heat and temperature, introduces specific heat capacity, and the heat equation. The script also discusses enthalpy of reaction, including exothermic and endothermic processes. It further explains concepts like enthalpy of formation and combustion, and uses Hess's Law to solve a practice problem, demonstrating how to calculate the enthalpy of formation for methane.
Takeaways
- 🔥 Thermochemistry involves the study of energy transfer in chemical reactions, particularly as heat.
- 🌡️ Temperature measures the motion of molecules, whereas heat is the transfer of energy.
- 💧 The bomb calorimeter is used to measure the total energy transferred to water molecules during an exothermic reaction.
- 🌡️ The Kelvin scale is used to measure temperature, and energy changes are measured in kilojoules (kJ) or calories.
- 🔥 Specific heat capacity (Cp) is defined as the energy required to heat 1 gram of a substance by 1 Kelvin.
- 💧 The heat equation (Q = mcΔT) relates the total energy transfer to the specific heat, mass, and temperature change of a substance.
- 🔄 Enthalpy of reaction (ΔH) measures the change in energy between reactants and products.
- 🔥 Exothermic reactions release energy, resulting in a negative ΔH, while endothermic reactions absorb energy, resulting in a positive ΔH.
- 🌐 Thermochemical equations show the relationship between the reactants, products, and energy changes in a reaction.
- 🔍 Hess's Law allows for the calculation of the enthalpy of formation of a compound by combining and manipulating known thermochemical equations.
- 🔥 The enthalpy of formation helps determine the stability of a compound, with more stable compounds having lower (more negative) values.
Q & A
What is thermochemistry?
-Thermochemistry is the study of energy transfer in chemical reactions, typically measured as heat.
What is the difference between heat and temperature?
-Temperature measures the motion of molecules, such as how fast water molecules oscillate in a bomb calorimeter, while heat is the transfer of energy, like the energy released during an exothermic reaction.
Why is the specific heat capacity important in thermochemistry?
-Specific heat capacity is crucial because it determines how much heat is required to change the temperature of a substance. It varies between materials, affecting how efficiently they can be heated or cooled.
What units are used to measure temperature and heat in the context of this script?
-Temperature is measured in degrees Kelvin (K), and heat is measured in joules (J).
How is the heat equation formulated?
-The heat equation is formulated as Q = mcΔT, where Q is the total energy transfer, m is the mass of the material, c is the specific heat capacity, and ΔT is the change in temperature.
What is enthalpy of reaction?
-Enthalpy of reaction, ΔH, represents the change in energy during a chemical reaction, which is the difference between the energy of the products and the energy of the reactants.
How does the enthalpy of reaction relate to the stability of a compound?
-A compound with a negative enthalpy of formation is more stable than its constituent elements because it releases energy when formed, indicating a lower energy state.
What is the difference between an exothermic and an endothermic reaction?
-An exothermic reaction releases energy (ΔH is negative), while an endothermic reaction absorbs energy (ΔH is positive).
What is the significance of Hess's Law in thermochemistry?
-Hess's Law allows the calculation of the enthalpy change for a reaction by combining known thermochemical equations, even if the reaction does not occur in one step.
How is the enthalpy of formation used to determine the stability of a compound?
-The enthalpy of formation is used to determine the stability of a compound by comparing its energy content to that of its constituent elements. A negative enthalpy of formation indicates a more stable compound.
What is the enthalpy of combustion and how is it measured?
-The enthalpy of combustion is the energy released when one mole of a substance is burned in excess oxygen. It is measured as the heat released per mole of reactant.
Outlines

此内容仅限付费用户访问。 请升级后访问。
立即升级Mindmap

此内容仅限付费用户访问。 请升级后访问。
立即升级Keywords

此内容仅限付费用户访问。 请升级后访问。
立即升级Highlights

此内容仅限付费用户访问。 请升级后访问。
立即升级Transcripts

此内容仅限付费用户访问。 请升级后访问。
立即升级浏览更多相关视频

CHM143 - Thermochemistry in our daily life
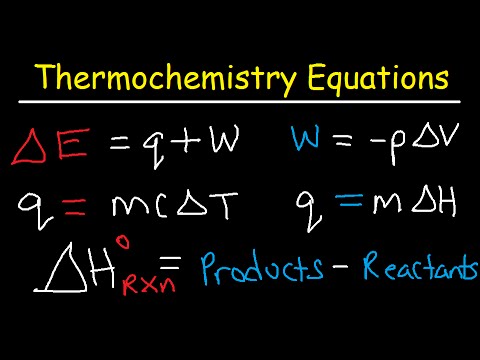
Thermochemistry Equations & Formulas - Lecture Review & Practice Problems

TERMOKIMIA - MATERI KIMIA KELAS 11 | Edcent.id

Termokimia (1) | Entalpi Dan Perubahan Entalpi | Persamaan Termokimia | Hukum Hess

REAKSI ENDOTERM & EKSOTERM - TERMOKIMIA

Termokimia • Part 5: Menentukan Entalpi Reaksi (Entalpi Pembentukan & Energi Ikatan)
5.0 / 5 (0 votes)
