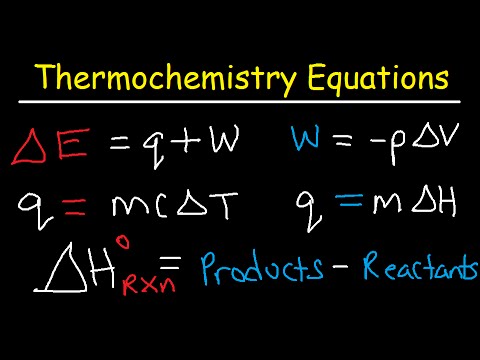CHM143 - Thermochemistry in our daily life
Summary
TLDRThis video introduces thermochemistry, the study of energy changes in chemical reactions and heat transfer. It covers historical developments, including the work of Lavoisier, Laplace, Hess, and others. The video explains how thermochemistry is applied in practical scenarios like calculating heat for phase changes, combustion reactions, and energy in vehicles. It also explores the importance of thermochemistry in renewable energy and power plants, as well as its role in refrigeration and air conditioning. The key takeaway is that thermochemistry helps determine the energy involved in chemical reactions, though it does not predict reaction speeds.
Takeaways
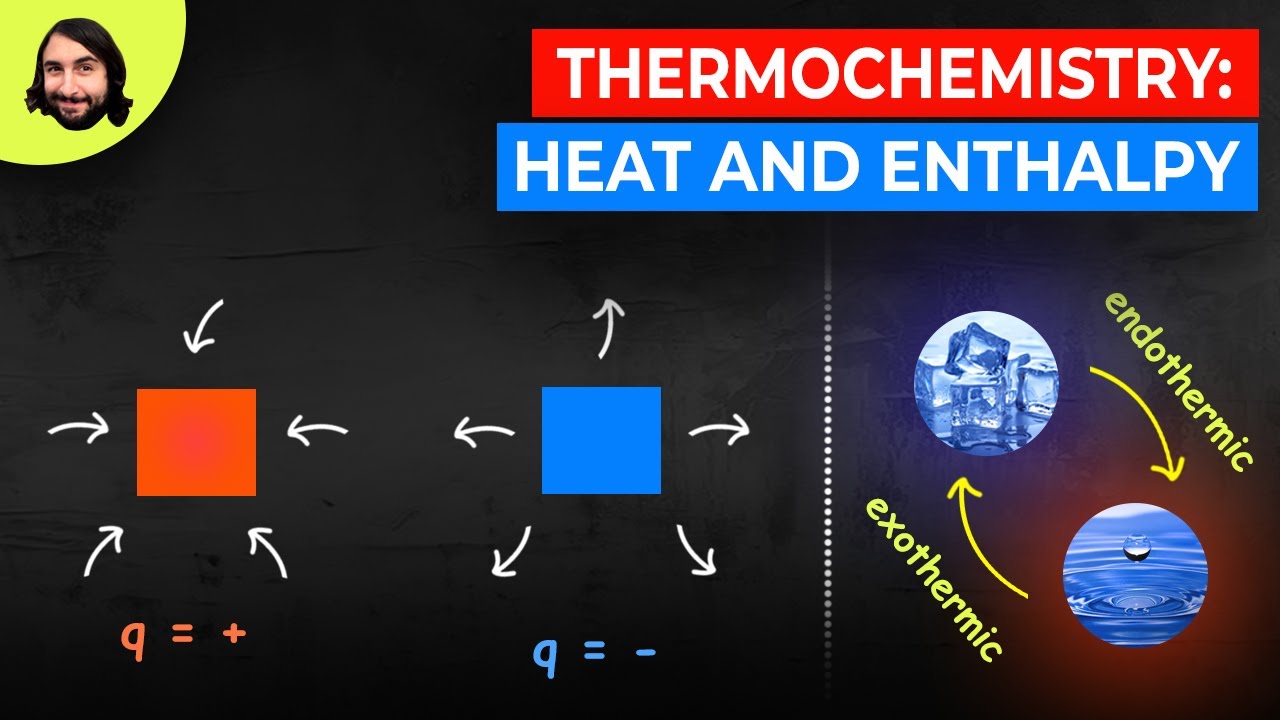
- 😀 Thermal chemistry studies energy changes during chemical reactions, focusing on heat energy movement.
- 😀 The history of thermal chemistry includes key contributions from Lavoisier and Laplace (1780s) about energy change in transformations.
- 😀 Hess' law (1840) states that energy change is the same regardless of how many steps a process takes, forming the basis for the First Law of Thermodynamics.
- 😀 Gibbs (1858) developed equations to calculate heat variations with temperature changes, aiding thermodynamic measurements.
- 😀 A calorimeter is used to measure heat changes in reactions by tracking temperature changes in an enclosed chamber.
- 😀 Thermochemistry helps calculate heat needed to raise the temperature of substances like water, calculate heat during phase changes, and understand heat evolution in chemical reactions.
- 😀 Examples from daily life, like vehicle engines (petrol, DC) and aircraft, are based on thermodynamic principles like the Carnot cycle.
- 😀 Thermodynamics is crucial for power plants (thermal, nuclear, hydroelectric) and renewable energy sources (solar, wind, geothermal).
- 😀 Refrigeration, air conditioning systems, and heat pumps work based on the second law of thermodynamics, which governs heat transfer.
- 😀 Thermochemistry is vital for predicting if a chemical reaction will occur and whether it will absorb or release energy.
- 😀 Thermochemistry helps assess the feasibility of chemical processes by calculating the energy involved, although it doesn’t predict the speed of reactions.
Q & A
What is thermal chemistry?
-Thermal chemistry is the study of energy changes that occur during chemical reactions, specifically the movement and transformation of heat energy.
Who were the key figures in the development of thermal chemistry?
-Key figures include La Russia and La Place, who introduced the idea that energy changes in any transformation are equal and opposite in reverse, and Hess, who formulated a law that energy change remains the same regardless of the reaction's steps.
What is Hess's Law, and why is it important?
-Hess's Law states that the energy change for a reaction is the same regardless of whether it occurs in one step or multiple. This principle is fundamental in formulating the First Law of Thermodynamics.
How does Gustav Kirchhoff's work contribute to thermal chemistry?
-Gustav Kirchhoff's work provided equations for the variation of heat, which are essential for calculating heat changes during temperature transitions in chemical reactions.
What is a calorimeter, and what is its purpose?
-A calorimeter is an enclosed chamber used to measure heat changes during chemical reactions. It helps determine fundamental units like heat capacity and the heat of combustion.
What are the primary modes of heat transfer discussed in the script?
-The primary modes of heat transfer are conduction, convection, and radiation.
How is thermochemistry applied in daily life?
-Thermochemistry is applied in everyday life through technologies like vehicle engines, refrigerators, air conditioning systems, and renewable energy power plants, all of which rely on the principles of heat transfer and energy changes.
What is the role of thermochemistry in energy generation?
-Thermochemistry plays a key role in the analysis of power plants, including thermal, nuclear, and renewable energy sources, by examining the heat energy conversion process and ensuring efficient energy production.
How do vehicles use thermochemistry in their engines?
-Vehicles use thermochemistry through internal combustion engines or other types of engines that rely on thermodynamic cycles, such as the Carnot cycle, to convert heat energy into mechanical work.
Why is thermochemistry important for environmental and economic sustainability?
-Thermochemistry helps evaluate the energy efficiency and environmental impact of chemical processes, determining the feasibility and economic viability of using particular reactions or energy systems in industries.
Outlines

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowMindmap

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowKeywords

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowHighlights

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowTranscripts

This section is available to paid users only. Please upgrade to access this part.
Upgrade Now5.0 / 5 (0 votes)