Quince minutos en la vida de un electrón
Summary
TLDRThis detailed script traces the evolution of our understanding of electrons, from early experiments with cathode rays to the development of quantum mechanics. It covers key discoveries such as the identification of the electron's negative charge, the early models of atomic structure, and the breakthroughs in the 20th century that led to the quantum theory. The script explores significant experiments by scientists like Millikan, Rutherford, and Einstein, ultimately culminating in the understanding of the wave-particle duality of electrons and the birth of quantum mechanics, reshaping our view of the atomic and subatomic world.
Takeaways
- 😀 The study of electrons and their behavior is crucial for understanding the structure of matter and chemical phenomena.
- 😀 Early experiments with discharge tubes revealed that elements emit colored light when energized, which can be analyzed into distinct spectral lines.
- 😀 The discovery of cathode rays by William Crookes in 1878 led to the identification of negatively charged particles, which were later called electrons.
- 😀 In 1897, J.J. Thomson demonstrated that cathode rays were composed of particles with negative electric charge, confirming the electron's existence.
- 😀 The photoelectric effect, discovered by Heinrich Hertz in 1887, revealed that light could eject electrons from a metal, further supporting the idea of discrete particles.
- 😀 Albert Einstein, in 1905, applied quantum theory to explain the photoelectric effect, showing that light consists of discrete energy packets called photons.
- 😀 Millikan's oil drop experiment in 1909 determined the fundamental charge of the electron, which helped define its mass and charge.
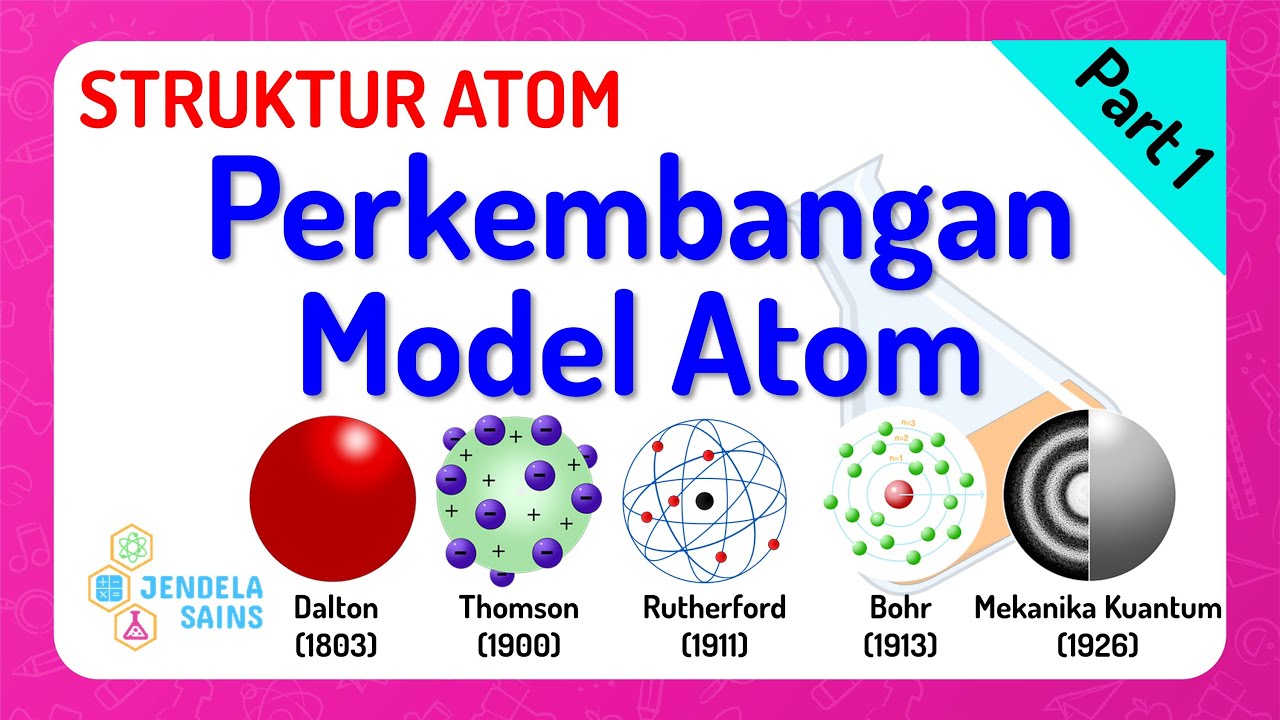
- 😀 Rutherford's gold foil experiment in 1911 showed that atoms have a tiny, dense nucleus containing positive charge, with electrons orbiting around it, forming a planetary model of the atom.
- 😀 Niels Bohr, in 1913, introduced a model of the atom where electrons move in fixed orbits without emitting radiation, explaining atomic spectra through quantization of angular momentum.
- 😀 In the 1920s, experiments by Clinton Davisson, Lester Germer, and others demonstrated the wave-like nature of electrons, confirming the wave-particle duality of matter.
- 😀 Quantum mechanics, developed by Heisenberg, Schrödinger, and others, provides a deeper understanding of electrons, replacing classical models with a probabilistic description of their behavior.
Q & A
What was the primary challenge in understanding the nature of electrons?
-The primary challenge was interpreting how electrons form molecules and contribute to chemical phenomena, which required understanding their behavior and properties in various experimental contexts.
How did scientists initially detect electrons?
-Electrons were first detected through the study of cathode rays in discharge tubes, where scientists observed a glowing beam when an electric potential was applied between electrodes in a vacuum.
What did the spectrum of light emitted by different elements reveal?
-The spectrum revealed that each element emitted light at specific wavelengths, which led to the discovery of atomic spectra and helped scientists identify the unique emission lines of each element.
What did William Crookes suggest about cathode rays in 1878?
-William Crookes suggested that cathode rays were made up of negatively charged particles that were repelled by the cathode, marking the first identification of electrons as particles.
How did J.J. Thomson contribute to the understanding of cathode rays?
-In 1897, J.J. Thomson conducted experiments with cathode rays and proved that they were negatively charged particles, which he later named electrons, demonstrating their role in electrical conduction.
What significant conclusion did Robert Millikan reach in 1909 regarding electrons?
-Robert Millikan determined the charge of an electron by conducting oil drop experiments, concluding that the charge was a fundamental constant, and allowed the calculation of the electron's mass.
What model did Ernest Rutherford propose for the structure of atoms in 1911?
-Ernest Rutherford proposed the planetary model of the atom, where a dense, positively charged nucleus is surrounded by electrons orbiting it, based on the results of his gold foil experiment.
What problem arose with Rutherford's model of the atom?
-Rutherford's planetary model couldn't explain atomic stability. According to classical electromagnetism, the electrons should emit radiation and spiral into the nucleus, leading to atomic instability, which wasn't observed.
How did Niels Bohr improve upon Rutherford's model in 1913?
-Niels Bohr introduced the idea of quantized orbits for electrons, suggesting that electrons could only occupy specific, stable orbits without radiating energy. This explanation resolved the stability issue in atoms.
What breakthrough did the double-slit experiment reveal about electrons?
-The double-slit experiment revealed that electrons exhibit both wave-like and particle-like properties. When not observed, they interfere with themselves like waves, but when measured, they behave as particles, confirming the wave-particle duality of matter.
Outlines

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowMindmap

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowKeywords

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowHighlights

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowTranscripts

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowBrowse More Related Video

Tudo se Transforma, História da Química, História dos Modelos Atômicos

Models of the Atom Timeline
Struktur Atom • Part 1: Perkembangan Model Atom

Uma Breve História do Átomo

2.4 - Estudo do Átomo - Modelo Atômico de Thomson (Modelo do Pudim de Passas)

Química - Natureza elétrica da matéria e núcleo atômico (prof. Luiz Landim)
5.0 / 5 (0 votes)