Rutherford Gold Foil Experiment - Backstage Science
Summary
TLDRThis video script recounts Rutherford's groundbreaking experiment using alpha particles to probe the atom's structure. By firing alpha particles through gold foil, he discovered that atoms are mostly empty space with a dense nucleus, contradicting the previous 'plum pudding' model. This experiment laid the foundation for modern particle physics and influenced technologies like the Large Hadron Collider, which continues to explore subatomic particles.
Takeaways
- 🔬 **Rutherford's Experiment**: The script describes Rutherford's experiment which aimed to understand the structure of the atom.
- 🧬 **Atom Structure**: Before Rutherford's experiment, atoms were thought to be a blob of positive charge with electrons embedded inside.
- 💥 **Alpha Particle Beam**: Rutherford used a beam of alpha particles, which are heavy, positively charged particles, to probe the atom's structure.
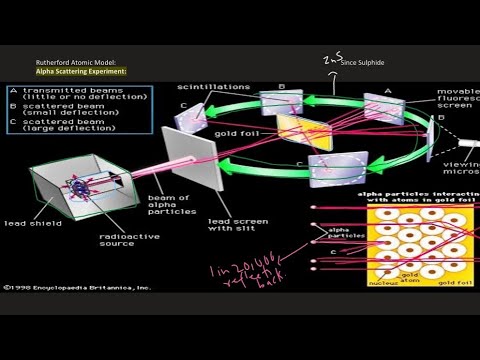
- 📍 **Gold Foil**: Alpha particles were directed through a very thin gold foil to observe their behavior.
- 🔎 **Detection Method**: Detectors were used to count particles going straight ahead and to measure scattering at different angles.
- 🕒 **Laborious Process**: The experiment involved sitting for hours in a dark room, observing a fluorescent screen for flashes of light caused by alpha particles.
- 📉 **Scattering Observations**: Most alpha particles passed straight through the gold foil, but some were deflected at various angles.
- 🌌 **Implications for Atom Structure**: The experiment indicated that atoms have a dense nucleus with mostly empty space, and electrons distributed around it.
- 🔬 **Birth of Particle Physics**: Rutherford's work marked the beginning of particle physics, which involves firing particle beams at targets to study their internal structure.
- 🌟 **Modern Parallels**: The script draws a parallel between Rutherford's experiment and modern particle physics, such as those conducted at the Large Hadron Collider.
- 📈 **Technological Advancement**: The experiment has evolved from a simple setup to the massive scale of the Large Hadron Collider, illustrating the progress in scientific technology.
Q & A
What was the primary purpose of Rutherford's gold foil experiment?
-The primary purpose of Rutherford's gold foil experiment was to test the existing model of atomic structure and to investigate the distribution of charge within the atom.
What were alpha particles and why were they significant in Rutherford's experiment?
-Alpha particles are heavy, positively charged particles, which are the nuclei of helium atoms. They were significant in Rutherford's experiment because their interaction with the gold foil helped reveal the presence of a small, dense, positively charged nucleus within the atom.
What was the thickness of the gold foil used in the experiment?
-The gold foil used in Rutherford's experiment was about 1.5 microns in thickness.
How did Rutherford and his colleagues detect the alpha particles in their experiment?
-Rutherford and his colleagues detected alpha particles by observing flashes of light on a tiny fluorescent screen in a dark room, which indicated the particles' presence.
What was the rate of alpha particle counts per second for the particles going straight ahead?
-The rate of alpha particle counts per second for the particles going straight ahead was between one and two thousand.
What did Rutherford deduce about the structure of the atom from the results of his experiment?
-Rutherford deduced that the atom had a heavy nucleus with mostly empty space, and electrons were distributed around it, which contradicted the then-prevailing 'plum pudding' model of the atom.
How did the experiment contribute to the field of particle physics?
-The experiment contributed to the field of particle physics by establishing a method of using particle scattering to deduce the internal structure of targets, which is a fundamental technique still used today.
What is the connection between Rutherford's experiment and the Large Hadron Collider?
-The connection is that both Rutherford's experiment and the Large Hadron Collider use the principle of firing a beam of particles at a target and observing the scattered particles to understand what is happening inside the target.
How has the scale of particle accelerators evolved from Rutherford's time to the present?
-The scale of particle accelerators has evolved significantly from Rutherford's tabletop experiment to the Large Hadron Collider, which has a circumference of 27 kilometers.
Why did most alpha particles pass through the gold foil without being deflected?
-Most alpha particles passed through the gold foil without being deflected because the nucleus of the gold atom is extremely small compared to the atom itself, leaving mostly empty space for the particles to pass through.
What was the significance of the occasional alpha particle bouncing back into the detector?
-The occasional alpha particle bouncing back into the detector was significant because it indicated a direct collision with the gold nucleus, providing evidence for the existence of a dense, positively charged center within the atom.
Outlines

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифMindmap

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифKeywords

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифHighlights

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифTranscripts

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифПосмотреть больше похожих видео
ALPHA PARTICLE SCATTERING EXPERIMENT || ATOMIC STRUCTURE || CHEMISTRY || CHAPTER 2 || LEVEL 11TH

2.5.1 - Descobrimento do núcleo atômico: Experimento de Rutherford

RutherfordÂs gold foil experiment | Electronic structure of atoms | Chemistry | Khan Academy

Rutherford's Experiment: Nuclear Atom

Rutherford's Gold Foil Experiment - Quick and Simple!

Rutherford’s Atomic Model - Part 1 | Atoms and Molecules | Infinity Learn
5.0 / 5 (0 votes)
