Hess's Law
Summary
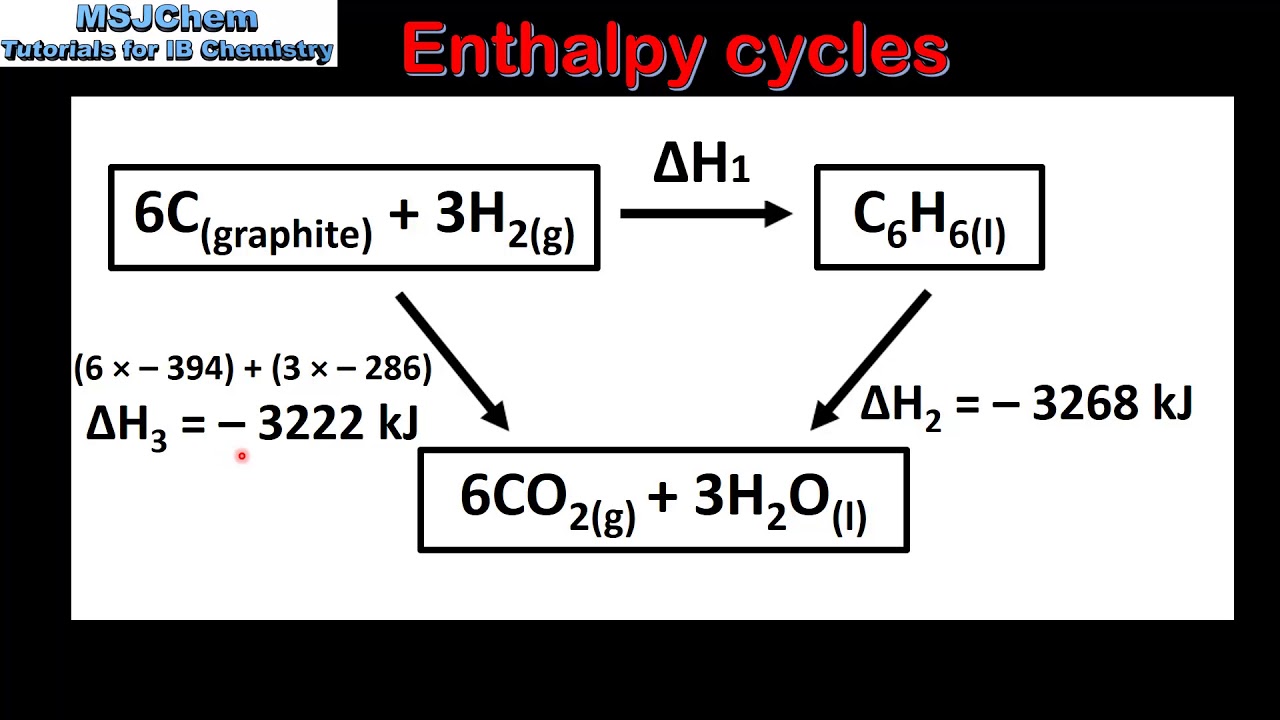
TLDRThis video explains Hess's Law and its application in determining the enthalpy change (Delta H) of a reaction when it's not directly available. The instructor demonstrates how to manipulate thermochemical equations to calculate Delta H by adding and adjusting other known reactions. The example provided involves calculating the Delta H for sulfur trioxide formation from sulfur and oxygen, showing how to reverse and scale reactions. The video concludes with a discussion of formation reactions, emphasizing the importance of having one mole of the product in such calculations.
Takeaways
- 📉 Hess's Law helps calculate the enthalpy change (ΔH) of a reaction by adding up the ΔH values of multiple related reactions.
- 📘 If ΔH for a reaction is unknown, you can either measure it using a calorimeter or use Hess’s Law with known reactions.
- 🔁 Hess's Law states that you can reverse or multiply chemical reactions to match the desired overall reaction.
- ⚖️ When multiplying a reaction, the ΔH value must also be multiplied by the same factor.
- ↔️ Reversing a reaction changes the sign of its ΔH; an exothermic reaction becomes endothermic and vice versa.
- 🧪 In an example with sulfur and oxygen forming sulfur trioxide, the reactions are manipulated to match the overall reaction.
- ✖️ Intermediate products, like sulfur dioxide in this case, can cancel out when added across reactions.
- 🔢 The total ΔH for the overall reaction is the sum of the ΔH values of the individual reactions.
- 📊 In formation reactions, it’s common to have fractional coefficients, which is acceptable as long as there’s one mole of product.
- 🌡️ Hess's Law is highly useful for calculating ΔH for reactions where direct experimental data is unavailable.
Q & A
What is the main method discussed in the video to calculate Delta H when it is not known?
-The video discusses using Hess's Law to calculate Delta H by manipulating other known reactions that add up to the overall desired reaction.
What is Hess's Law?
-Hess's Law states that it is possible to add two or more thermochemical equations to produce a final equation for a reaction, and the sum of the enthalpy changes for the individual reactions is the enthalpy change for the final reaction.
What example reaction is used in the video to explain Hess's Law?
-The reaction used is sulfur (S) plus oxygen gas (O2) yielding sulfur trioxide (SO3), where the Delta H of the reaction is initially unknown.
How is the first reaction modified to match the desired reaction?
-The first reaction, which has one mole of sulfur (S), is multiplied by 2 to have two moles of sulfur, matching the reactant side of the target reaction.
What happens to the Delta H when the reaction is multiplied by 2?
-The Delta H is also multiplied by 2. For example, if the original Delta H was -297 kJ, it becomes -594 kJ after multiplying the reaction by 2.
Why is the second reaction flipped in the explanation?
-The second reaction is flipped because sulfur dioxide (SO2) is on the product side, but it is needed on the reactant side to match the desired reaction. Reversing the reaction also changes the sign of the Delta H.
How do the sulfur dioxide (SO2) molecules get canceled out?
-The two moles of sulfur dioxide (SO2) on the reactant side of the flipped reaction cancel out the two moles of sulfur dioxide on the product side of the first reaction.
What is the final Delta H for the overall reaction after using Hess's Law?
-The final Delta H for the reaction is -792 kJ after adding the modified Delta H values from the two reactions.
Why is the equation divided by 2 at the end?
-The equation is divided by 2 to convert it into a formation reaction, which involves forming one mole of the product, sulfur trioxide (SO3), from its elements. Formation reactions require one mole of the product.
What is the final Delta H per mole for the formation reaction?
-After dividing by 2, the final Delta H for the formation of one mole of sulfur trioxide (SO3) is -396 kJ per mole.
Outlines

Esta sección está disponible solo para usuarios con suscripción. Por favor, mejora tu plan para acceder a esta parte.
Mejorar ahoraMindmap

Esta sección está disponible solo para usuarios con suscripción. Por favor, mejora tu plan para acceder a esta parte.
Mejorar ahoraKeywords

Esta sección está disponible solo para usuarios con suscripción. Por favor, mejora tu plan para acceder a esta parte.
Mejorar ahoraHighlights

Esta sección está disponible solo para usuarios con suscripción. Por favor, mejora tu plan para acceder a esta parte.
Mejorar ahoraTranscripts

Esta sección está disponible solo para usuarios con suscripción. Por favor, mejora tu plan para acceder a esta parte.
Mejorar ahoraVer Más Videos Relacionados
5.0 / 5 (0 votes)