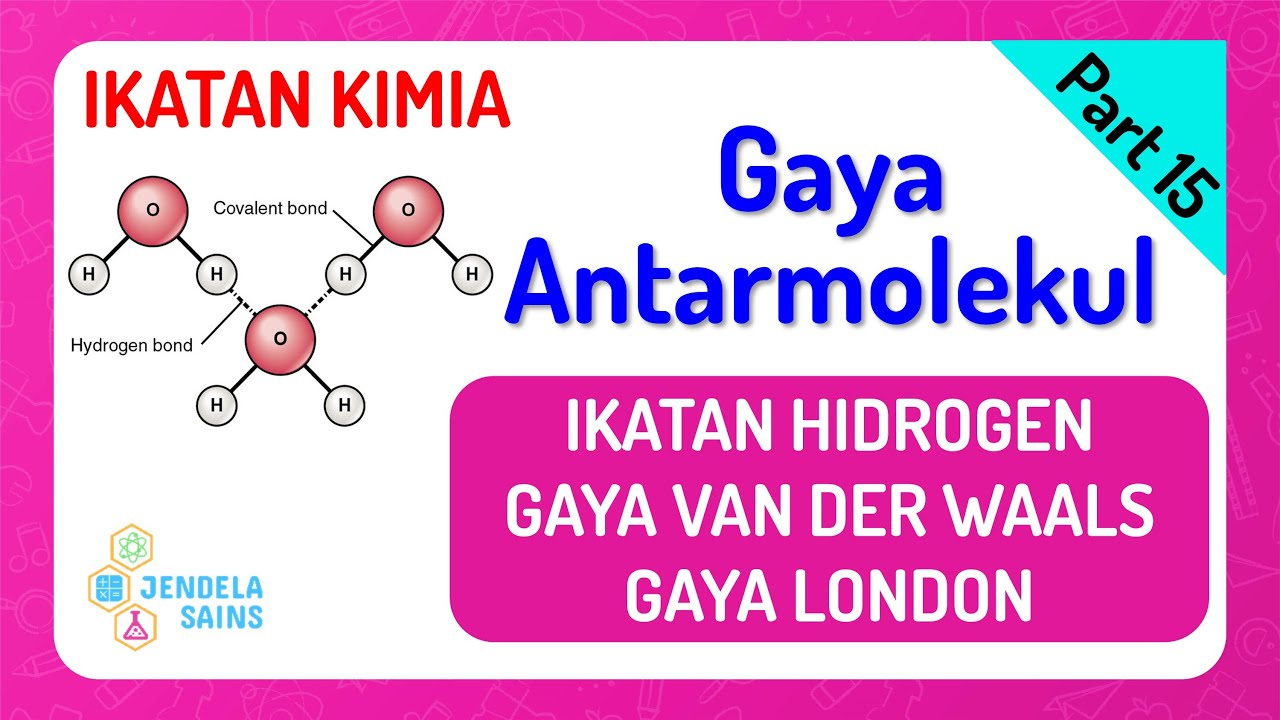
Hydrogen Bonding | Chemistry
Summary
TLDRThis lecture explores hydrogen bonding, focusing on its nature as an intermolecular force rather than a chemical bond. It explains how hydrogen bonding occurs in highly polar molecules like hydrogen fluoride, where the electronegative fluorine pulls electrons towards itself, creating a partial positive charge on hydrogen. The lecture clarifies that hydrogen bonds are not present in all hydrogen compounds, using methane, ethane, and propane as examples due to their nonpolar C-H bonds. It introduces the 'NORTH' mnemonic for elements that form hydrogen bonds when bonded with hydrogen, specifically nitrogen, oxygen, and fluorine. The lecture also compares hydrogen bonds to other intermolecular forces, highlighting their relative strengths and uses the example of water's high boiling point to illustrate the impact of hydrogen bonding on physical properties.
Takeaways
- 📘 Hydrogen bonding is an intermolecular force between molecules, not a chemical bond.
- ⚛️ In a hydrogen fluoride (HF) molecule, fluorine is much more electronegative than hydrogen, leading to a polar bond with partial charges.
- ⚡ The electronegativity difference between hydrogen and fluorine in HF is 1.9, making HF highly polarized.
- 🧲 Hydrogen bonding occurs when the negative pole of one HF molecule attracts the positive pole of another HF molecule.
- 🌊 Hydrogen bonds are intermolecular forces between highly polarized molecules, such as HF, NH₃ (ammonia), and H₂O (water).
- ❌ Hydrogen bonds do not exist in all hydrogen compounds; they are absent in nonpolar molecules like methane, ethane, and propane.
- 🧠 Use the mnemonic 'NORTH' (Nitrogen, Oxygen, Fluorine) to remember that hydrogen bonds form with these elements.
- 🔬 Hydrogen bonds are a special type of dipole-dipole interaction and are generally stronger than other dipole-dipole forces but much weaker than covalent bonds.
- 🌡️ Water (H₂O) has a higher boiling point than hydrogen sulfide (H₂S) because water contains hydrogen bonds, while H₂S does not.
- 📊 Hydrogen bonds are significantly weaker than covalent bonds, approximately 20 times weaker.
Q & A
What is hydrogen bonding?
-Hydrogen bonding is an attractive force between highly polarized molecules, such as hydrogen fluoride, ammonia, and water. It is an intermolecular force and not a chemical bond.
Why does hydrogen fluoride have hydrogen bonding?
-Hydrogen fluoride has hydrogen bonding because fluorine is highly electronegative, creating a large difference in electronegativity with hydrogen. This leads to partial charges and an attraction between molecules.
Does hydrogen bonding exist in all hydrogen compounds?
-No, hydrogen bonding does not exist in all hydrogen compounds. It only occurs in compounds where hydrogen is bonded to highly electronegative elements like nitrogen, oxygen, or fluorine.
Why doesn’t hydrogen bonding occur in compounds like methane, ethane, and propane?
-Hydrogen bonding doesn’t occur in these compounds because the difference in electronegativity between carbon and hydrogen is too small, making the bonds non-polar and unable to form hydrogen bonds.
Which elements form hydrogen bonds with hydrogen?
-Hydrogen forms hydrogen bonds with nitrogen, oxygen, and fluorine. A helpful mnemonic to remember this is 'north' – nitrogen, oxygen, and fluorine.
What is the difference between hydrogen bonds and covalent bonds?
-Hydrogen bonds are intermolecular forces between molecules, while covalent bonds are intramolecular forces within atoms. Covalent bonds are 20 times stronger than hydrogen bonds.
Why does water have a higher boiling point than hydrogen sulfide?
-Water has a higher boiling point than hydrogen sulfide because water contains hydrogen bonding, which requires more energy to break, while hydrogen sulfide lacks hydrogen bonding.
Is hydrogen bonding stronger than dipole-dipole forces?
-Yes, hydrogen bonding is a special type of dipole-dipole force and is about 10 times stronger than typical dipole-dipole interactions.
Why is hydrogen bonding important in determining the properties of a substance?
-Hydrogen bonding significantly affects properties like boiling points, melting points, and solubility by increasing intermolecular attractions, as seen in water’s high boiling point.
In which common substances does hydrogen bonding occur?
-Hydrogen bonding occurs in ammonia (NH3), water (H2O), and hydrogen fluoride (HF), where hydrogen is covalently bonded to nitrogen, oxygen, or fluorine.
Outlines

Esta sección está disponible solo para usuarios con suscripción. Por favor, mejora tu plan para acceder a esta parte.
Mejorar ahoraMindmap

Esta sección está disponible solo para usuarios con suscripción. Por favor, mejora tu plan para acceder a esta parte.
Mejorar ahoraKeywords

Esta sección está disponible solo para usuarios con suscripción. Por favor, mejora tu plan para acceder a esta parte.
Mejorar ahoraHighlights

Esta sección está disponible solo para usuarios con suscripción. Por favor, mejora tu plan para acceder a esta parte.
Mejorar ahoraTranscripts

Esta sección está disponible solo para usuarios con suscripción. Por favor, mejora tu plan para acceder a esta parte.
Mejorar ahoraVer Más Videos Relacionados

PART 1 IKATAN KIMIA DAN GAYA ANTARMOLEKUL
Ikatan Kimia • Part 15: Gaya Antar Molekul: Ikatan Hidrogen & van der Waals, Gaya London

Ikatan Kimia (9) | Gaya Antar Molekul | Ikatan Antar Molekul |Kimia kelas 10

Class 11 Chemistry | Chemical Bonding and Molecular Str. in 15 Minutes | Rapid Revision by BP Sir

Intermolecular Forces | Chemistry

Grade 11 Chemistry: Chemical bonding Atomic Combinations
5.0 / 5 (0 votes)
