Stoikiometri Senyawa • Part 1: Kadar Zat - Persen Massa, Persen Volume, bpj / ppm
Summary
TLDRThis educational video from Jendela Sains explores stoichiometry and the concept of substance concentration in high school chemistry. It explains three key ways to express concentration: mass percent, volume percent, and parts per million (PPM), detailing their formulas and applications. The video provides step-by-step examples, calculating the mass of solutes in solutions, maximum allowable concentrations in drinking water, and molarity of concentrated sulfuric acid. Practical tips and a quick formula for molarity are included to simplify calculations, making complex stoichiometry concepts accessible and engaging for students seeking to master the fundamentals of chemical concentrations.
Takeaways
- 😀 Kadar zat refers to the concentration of a substance in a sample, which can be expressed using different methods such as mass percent, volume percent, and parts per million (PPM).
- 😀 Mass percent (%m) measures the mass of a substance in 100 grams of a sample, and the formula is: (Mass of Substance / Mass of Sample) * 100%.
- 😀 Volume percent (%v) calculates the volume of a substance in 100 ml of a solution, often used for liquid solutions or solutes.
- 😀 Parts per million (PPM) expresses the concentration of a substance as mass or volume per one million parts of the sample.
- 😀 The formula for PPM is: (Mass of Substance / Mass of Sample) * 10^6 or (Volume of Substance / Volume of Sample) * 10^6.
- 😀 Mass percent and volume percent are commonly used in chemistry to determine the concentration of substances in mixtures and solutions.
- 😀 PPM is useful when measuring extremely small concentrations, such as pollutants in water or air.
- 😀 In a sample of limestone, if 100 grams contain 70 grams of calcium carbonate, the mass percent of calcium carbonate is 70%.
- 😀 A sample problem showed how to calculate the mass of a solute (CH₃COOH) in a solution with known concentration, volume, and density using mass percent.
- 😀 The video also demonstrated how to calculate the maximum allowable concentration of a substance, such as iron in drinking water, using PPM and density.
- 😀 Molarity and molality are related concepts that also measure concentration, and can be calculated by converting units like volume into liters or using known formulas.
Q & A
What is meant by 'kadar zat' in chemistry?
-'Kadar zat' refers to the concentration or amount of a substance in a mixture or solution. It can be expressed in terms of mass percent, volume percent, parts per million (ppm), or molarity.
How is mass percent (% m/m) calculated?
-Mass percent is calculated by dividing the mass of the solute by the total mass of the sample and multiplying by 100%: % m/m = (mass of solute / mass of sample) × 100%.
What is the difference between percent mass and percent volume?
-Percent mass (% m/m) measures the mass of the solute relative to the total mass of the sample, while percent volume (% v/v) measures the volume of the solute relative to the total volume of the solution, commonly used for liquid mixtures.
How do you calculate parts per million (ppm) for a substance?
-PPM is calculated as the mass of the substance divided by the mass of the sample, multiplied by one million: ppm = (mass of substance / mass of sample) × 10^6. It can also be expressed based on volume if appropriate.
How can you determine the mass of a solute in a solution given percent mass and solution volume?
-First, calculate the mass of the solution using its volume and density (mass = density × volume). Then apply the mass percent formula: mass of solute = % m/m × mass of solution / 100%.
What is the relationship between molarity, mass of solute, and volume of solution?
-Molarity (M) is the number of moles of solute divided by the volume of the solution in liters: M = n / V. If the solute's mass is known, the number of moles n can be calculated as n = mass / molar mass.
How do you calculate molarity if you only know the solution's density and percent mass?
-Use the formula M = (density × 10 × % mass) / molar mass. This converts density and percent mass into moles per liter of solution efficiently.
In the given example, how much CH3COOH is dissolved in 200 ml of 25% m/m solution with a density of 1.05 g/ml?
-The mass of the solution is 200 ml × 1.05 g/ml = 210 g. The mass of CH3COOH is then (25/100) × 210 g = 52.5 g.
What is the maximum mass of iron allowed in a 200 ml glass of drinking water with 0.28 ppm Fe?
-Mass of water = 200 g. Mass of Fe = (0.28 / 10^6) × 200 g = 5.6 × 10^-5 g. This is the maximum allowed iron content.
How do you calculate molarity for concentrated H2SO4 given 98% mass and density 1.8 g/ml?
-Assume 100 g solution: mass of H2SO4 = 98 g. Volume of solution = 100 g / 1.8 g/ml ≈ 55.6 ml = 0.0556 L. Moles of H2SO4 = 98 g / 98 g/mol = 1 mol. Molarity = 1 mol / 0.0556 L ≈ 18 M.
Outlines

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowMindmap

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowKeywords

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowHighlights

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowTranscripts

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowBrowse More Related Video

Fluida Statis • Part 7: Viskositas, Hukum Stokes, Kecepatan Terminal

Hakikat Ilmu Kimia Kelas 10 • Part 1: Hakikat dan Peran Ilmu Kimia dalam Kehidupan
KONSEP MOL - KIMIA - MATERI UTBK SBMPTN DAN SIMAK UI - UTBK 2022 | SIMAK UI 2022

Concentration of solutions Chemistry
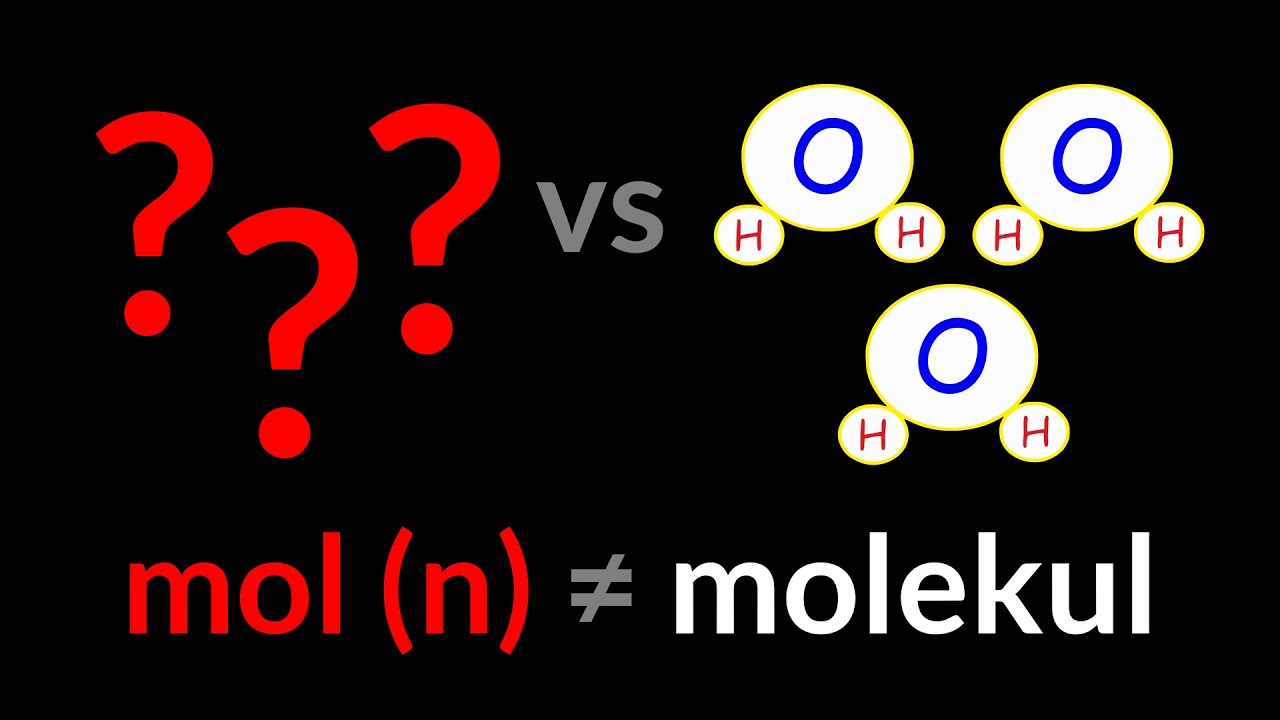
Stoikiometri 1 (mol adalah jumlah)

Matriks Matematika Kelas 11 • Part 23: Menyelesaikan SPLTV dengan Metode Determinan Matriks
5.0 / 5 (0 votes)