VSEPR Theory - Basic Introduction
Summary
TLDRThis video explains VSEPR (Valence Shell Electron Pair Repulsion) theory and its application in predicting molecular geometry. It covers key shapes including linear, trigonal planar, tetrahedral, trigonal pyramidal, and bent geometries, highlighting their bond angles and central atom arrangements. Using examples like BeCl₂, CO₂, BH₃, CH₄, NH₃, H₂O, and SO₂, the video illustrates how lone pairs influence molecular shapes and angles. It also provides practical tips, such as recognizing patterns based on element groups, helping viewers understand how electrons repel to create three-dimensional molecular structures. This guide simplifies complex concepts for easy learning and exam preparation.
Takeaways
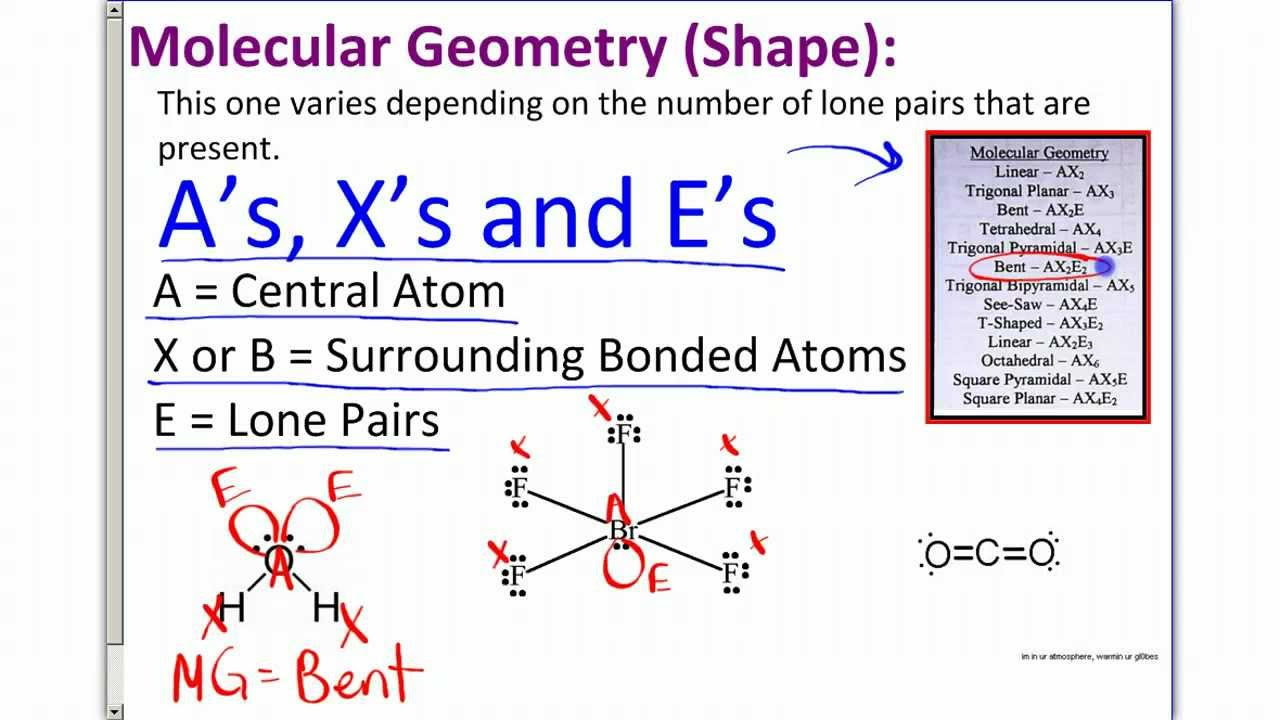
- 😀 VSEPR theory (Valence Shell Electron Pair Repulsion) predicts molecular geometry based on electron pair repulsion.
- 😀 Electrons repel each other and arrange themselves as far apart as possible, determining the shape of molecules.
- 😀 Linear molecular geometry occurs when a central atom is bonded to two atoms, with a bond angle of 180° (e.g., BeCl₂, CO₂, I₃⁻).
- 😀 Trigonal planar geometry has a central atom bonded to three atoms with no lone pairs, forming a flat shape with bond angles of 120° (e.g., BH₃, COCl₂, AlCl₃).
- 😀 Tetrahedral geometry has a central atom bonded to four atoms with bond angles of approximately 109.5° (e.g., CH₄, SiF₄).
- 😀 Trigonal pyramidal geometry has a central atom bonded to three atoms and one lone pair, slightly reducing the bond angle to ~107° (e.g., NH₃, PH₃, AsH₃).
- 😀 Bent molecular geometry occurs when a central atom is bonded to two atoms with one or two lone pairs, reducing the bond angle to ~104.5° (e.g., H₂O, SO₂).
- 😀 Lone pairs reduce bond angles because they repel bonded electrons more strongly, affecting molecular shape.
- 😀 Tetrahedral → Trigonal Pyramidal → Bent illustrates how replacing bonded atoms with lone pairs decreases bond angles gradually.
- 😀 Trigonal planar vs. trigonal pyramidal: trigonal planar has no lone pairs on the central atom, while trigonal pyramidal has one lone pair, altering the geometry.
- 😀 Group trends: molecules with trigonal planar geometry often involve group 3A elements, while trigonal pyramidal molecules often involve group 5A elements.
Q & A
What does VSEPR stand for and what is its main principle?
-VSEPR stands for Valence Shell Electron Pair Repulsion. Its main principle is that electron pairs around a central atom repel each other and will arrange themselves as far apart as possible to minimize repulsion, which helps predict the shape of molecules.
What is the bond angle in a linear molecular geometry and can you give examples?
-The bond angle in a linear molecular geometry is 180 degrees. Examples include BeCl2, CO2, and the triiodide ion (I3-).
Describe the trigonal planar structure and its typical bond angle.
-A trigonal planar structure has a central atom bonded to three other atoms with no lone pairs on the central atom. The atoms are spaced evenly in a flat plane with a bond angle of approximately 120 degrees. Examples include BH3 and COCl2.
What characterizes a tetrahedral molecular geometry and what is its bond angle?
-A tetrahedral geometry has a central atom bonded to four other atoms in a three-dimensional arrangement, with a bond angle of about 109.5 degrees. Examples include methane (CH4) and silicon tetrafluoride (SiF4).
How does a trigonal pyramidal structure differ from a trigonal planar structure?
-A trigonal pyramidal structure has a central atom with three bonded atoms and one lone pair, giving it a three-dimensional shape, while a trigonal planar structure has three bonded atoms and no lone pairs, forming a flat plane. Example of trigonal pyramidal: NH3.
What is the bond angle in ammonia (NH3) and why is it different from tetrahedral?
-The bond angle in ammonia is about 107 degrees. It is slightly less than the tetrahedral angle of 109.5 degrees due to the lone pair on nitrogen, which exerts greater repulsion and slightly compresses the H-N-H angles.
Explain the bent molecular geometry and provide examples with bond angles.
-Bent molecular geometry occurs when a central atom has two bonded atoms and one or more lone pairs, resulting in a non-linear shape. Examples include water (H2O) with a bond angle of 104.5 degrees and sulfur dioxide (SO2) with a bond angle slightly less than 120 degrees.
How does the presence of lone pairs affect bond angles in molecules?
-Lone pairs occupy more space than bonded atoms because of their stronger repulsion, which pushes bonded atoms closer together and reduces the bond angles compared to ideal geometries.
Which groups in the periodic table are commonly associated with trigonal planar and trigonal pyramidal structures?
-Elements in Group 3A often form trigonal planar structures (e.g., BH3, AlCl3), while elements in Group 5A typically form trigonal pyramidal structures (e.g., NH3, PH3, AsH3) when bonded to hydrogen atoms.
How are tetrahedral, trigonal pyramidal, and bent structures related in terms of electron groups?
-All three structures are derived from four electron groups around the central atom. Tetrahedral has four bonded atoms, trigonal pyramidal has three bonded atoms and one lone pair, and bent has two bonded atoms and two lone pairs. Lone pairs reduce bond angles slightly compared to the ideal tetrahedral angle of 109.5 degrees.
Why is SO2's bond angle slightly less than 120 degrees despite resembling a trigonal planar structure?
-SO2 has two bonded atoms and one lone pair on the sulfur atom. The lone pair occupies more space, causing the bond angle to be slightly less than the ideal 120 degrees of a trigonal planar structure.
How can you quickly distinguish between trigonal planar and trigonal pyramidal structures in simple molecules?
-You can distinguish them by checking the central atom's group in the periodic table and the presence of lone pairs. Group 3A elements without lone pairs typically form trigonal planar structures, while Group 5A elements with one lone pair typically form trigonal pyramidal structures.
Outlines

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowMindmap

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowKeywords

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowHighlights

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowTranscripts

This section is available to paid users only. Please upgrade to access this part.
Upgrade Now5.0 / 5 (0 votes)