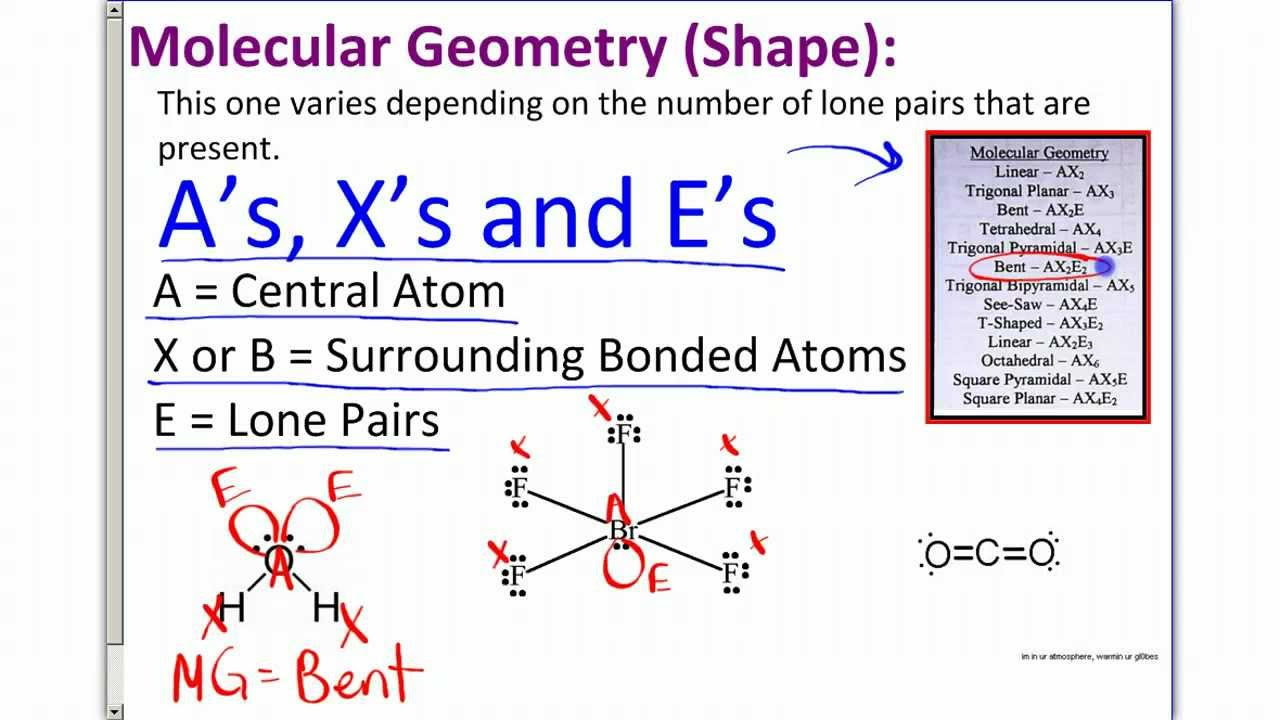
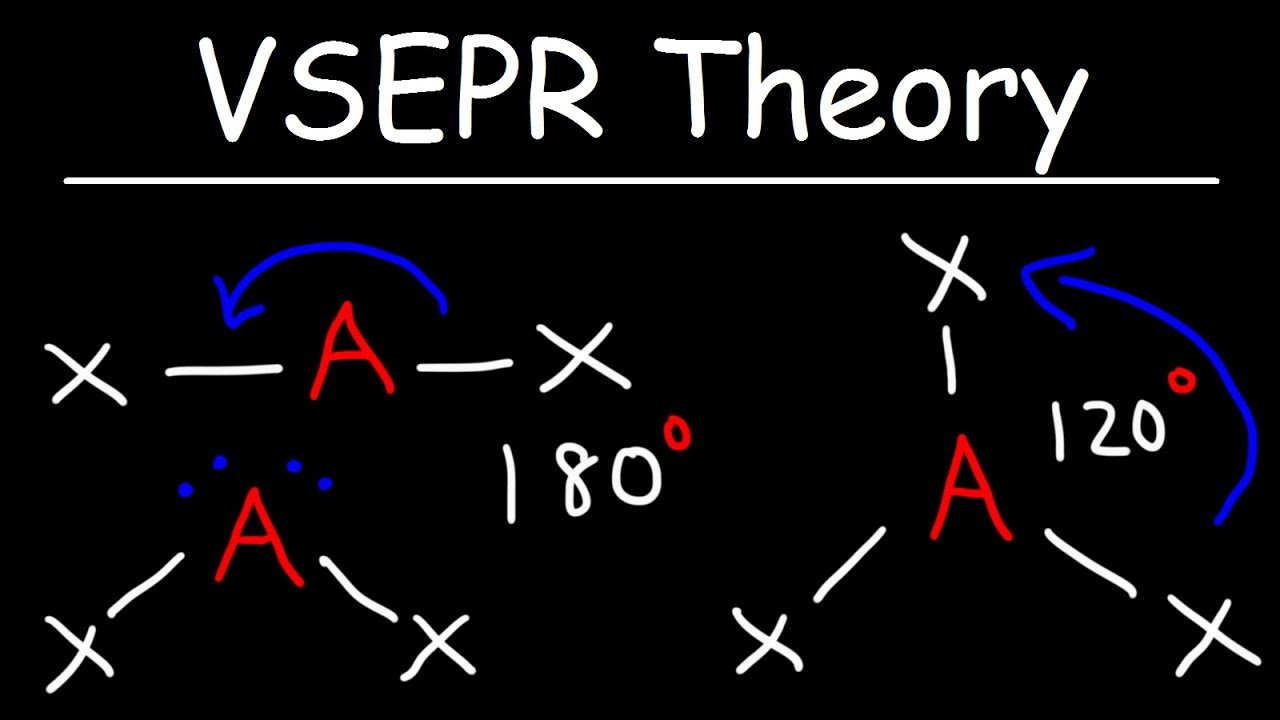
VSEPR Theory
Summary
TLDRThe script introduces the Valence Shell Electron Pair Repulsion (VSEPR) theory, a model used to predict molecular geometry in three dimensions. It explains how electron pairs around a central atom repel each other, influencing molecular shape. The theory covers geometries from linear (two regions) to tetrahedral (four regions), and extends to trigonal bipyramidal (five regions) and octahedral (six regions). These shapes are crucial for understanding organic chemistry and molecular behavior.
Takeaways
- 📚 The Valence Shell Electron Pair Repulsion (VSEPR) theory is used to predict the 3D shapes of molecules based on the repulsion between electron pairs around a central atom.
- 🔬 VSEPR theory suggests that electron regions around a central atom will arrange themselves to be as far apart as possible due to their negative charge.
- 📏 With two electron regions, a molecule adopts a linear shape with a bond angle of 180 degrees.
- 🍕 Three electron regions result in a trigonal planar shape with bond angles of 120 degrees each.
- 🔲 Four electron regions create a tetrahedral geometry, with bond angles of approximately 109.5 degrees.
- 📐 The bond angles decrease as more electron regions are added, due to increased repulsion among the electron pairs.
- 🔺 With five electron regions, a molecule forms a trigonal bipyramidal shape, combining linear and trigonal planar geometries.
- 🔶 Six electron regions result in an octahedral geometry, resembling the XYZ coordinate system with bond angles of 90 or 180 degrees.
- 🧩 VSEPR theory accounts for molecules with expanded octets, which can have five or six electron regions, leading to more complex 3D shapes.
- 🌐 The theory is fundamental to understanding molecular geometry, particularly in organic chemistry, and helps predict the spatial arrangement of atoms in molecules.
Q & A
What is the Valence Shell Electron Pair Repulsion (VSEPR) theory?
-VSEPR theory is a model used to predict the shapes of molecules by considering the repulsion between electron pairs in the valence shell of the central atom.
Why is it necessary to consider 3-dimensional molecular shapes?
-Molecules exist in a three-dimensional world, and considering their 3D shapes helps to understand their real-life behavior and interactions more accurately.
How does the VSEPR theory work?
-The VSEPR theory works by counting electron regions around the central atom and predicting the molecular shape based on the repulsion between these negatively charged regions.
What is the molecular shape when there are two electron regions?
-When there are two electron regions, the molecule adopts a linear shape with a bond angle of 180 degrees.
What is the molecular shape when there are three electron regions?
-With three electron regions, the molecule forms a trigonal planar shape with bond angles of 120 degrees.
What is the tetrahedral geometry and how many regions does it have?
-Tetrahedral geometry is a molecular shape with four regions around the central atom, characterized by bond angles of 109.5 degrees.
What is the significance of the bond angles getting smaller as more regions are added?
-As more electron regions are added, the bond angles decrease due to increased repulsion between the electron pairs, leading to more compact molecular shapes.
What is a trigonal bipyramidal shape and how is it formed?
-A trigonal bipyramidal shape is formed when there are five electron regions, consisting of two linear regions and three regions in a trigonal planar arrangement perpendicular to the linear ones.
How does the octahedral geometry relate to the XYZ coordinate system?
-An octahedral geometry is similar to the XYZ coordinate system, with six regions positioned at 90 or 180-degree angles, resembling the axes and coordinate points in three-dimensional space.
What is the total number of bond angles in an octahedral geometry?
-In an octahedral geometry, there are eight bond angles, with some being 90 degrees and others 180 degrees, forming a closed three-dimensional figure with eight sides.
How many electronic geometries are there in total according to the VSEPR theory?
-According to the VSEPR theory, there are five electronic geometries around the central atom: linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral.
Outlines

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowMindmap

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowKeywords

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowHighlights

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowTranscripts

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowBrowse More Related Video
5.0 / 5 (0 votes)