Simple Distillation | #aumsum #kids #science #education #children
Summary
TLDRSimple Distillation is a purification technique that separates liquid mixtures by heating to vaporize components with different boiling points. The process involves heating a mixture of acetone and water, monitoring the temperature, and using a condenser to collect the vaporized acetone at 56°C, leaving water behind at 100°C. This method is effective when boiling points differ significantly but is less efficient for mixtures with boiling points less than 25°C apart, prompting the use of fractional distillation for more precise separation.
Takeaways
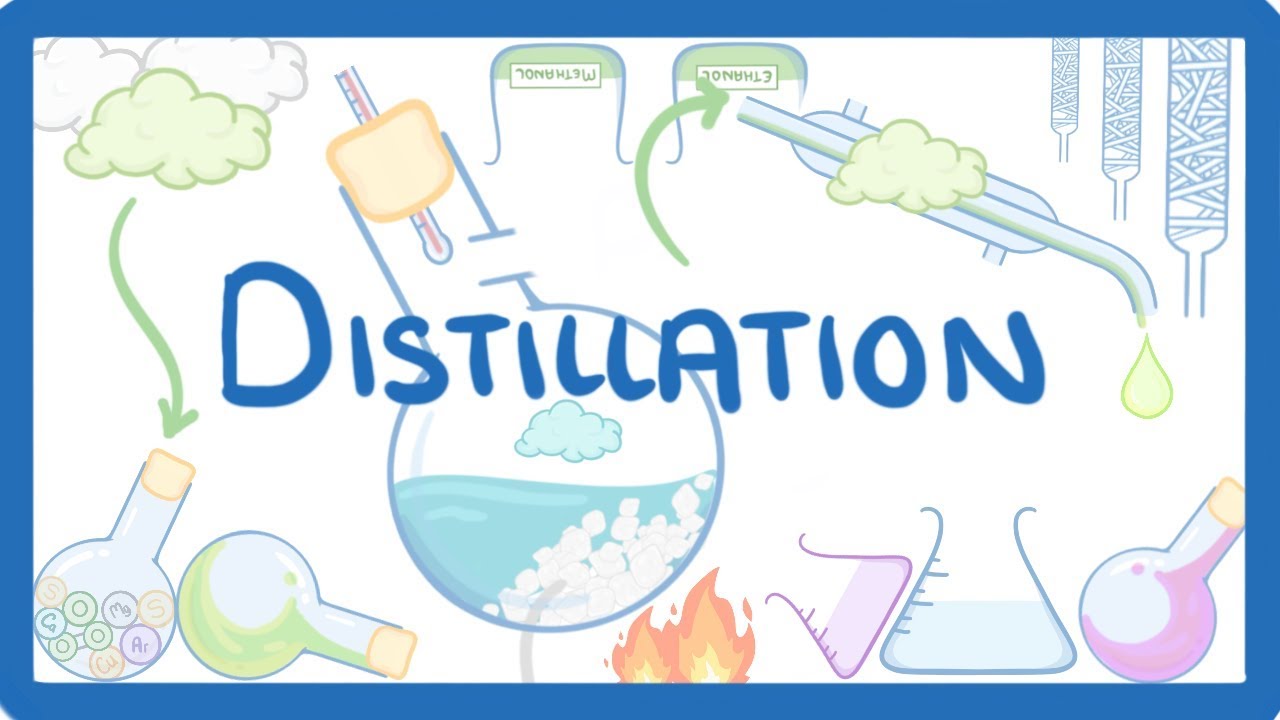
- 🔬 Simple Distillation is the process of heating a liquid mixture to form vapor and then cooling that vapor to get a liquid.
- 🧪 The purpose of distillation is to purify a liquid by separating its components.
- 🌡️ An activity demonstrates the process using a mixture of acetone and water in a distillation flask with a thermometer.
- 💧 A water condenser is used to cool the vapors, with cold water running through its jacket.
- 🍶 A beaker is placed at the outlet of the condenser to collect the condensed liquid.
- 🔥 Heating the mixture causes acetone to vaporize at 56 degrees Celsius, while water remains.
- 🌀 The vapors from acetone condense in the condenser and are collected in the beaker.
- 🚫 Simple distillation is not effective when the boiling point difference between two components is less than 25 degrees Celsius.
- ♻️ Repeating the process can improve separation, but it is time-consuming.
- 🔄 Fractional distillation is a more efficient method for separating components with close boiling points.
Q & A
What is the process of simple distillation?
-Simple distillation is the process of heating a liquid mixture to form vapor and then cooling that vapor to obtain a liquid, used to purify a liquid by separating its components.
Why is a thermometer used in simple distillation?
-A thermometer is used to monitor the temperature of the mixture during distillation, which helps to determine when a specific component, like acetone at 56 degrees Celsius, starts to vaporize.
What is the purpose of a water condenser in distillation?
-A water condenser is used to cool the vapors produced during distillation, causing them to condense back into a liquid, which is then collected separately.
How does the condenser keep the temperature cool during distillation?
-The condenser has cold water running through its jacket, which helps to maintain a cool temperature and facilitate the condensation of vapors.
What is collected in the beaker during the distillation of acetone and water?
-During the distillation of acetone and water, the condensed acetone is collected in the beaker, while the water remains in the flask.
Why does acetone start to vaporize at 56 degrees Celsius?
-Acetone has a boiling point of 56 degrees Celsius, so it starts to vaporize at this temperature, allowing it to be separated from water, which has a higher boiling point.
What is the boiling point of water and how does it relate to distillation?
-The boiling point of water is 100 degrees Celsius. This is significant in distillation because it is higher than that of acetone, allowing acetone to be separated first when the mixture is heated.
Why is there a limitation on the difference between boiling points for simple distillation to work effectively?
-Simple distillation works best when there is a significant difference between the boiling points of the components in the mixture. If the difference is less than 25 degrees Celsius, the components may not separate and purify completely.
What is the alternative to simple distillation when the boiling points of components are too close together?
-Fractional distillation is a special type of distillation used when the boiling points of components are too close together, allowing for more effective separation and purification.
Why might one need to repeat the simple distillation process multiple times?
-Repeating the simple distillation process multiple times can help to achieve a higher level of separation and purity of the components, although it can be time-consuming.
How does fractional distillation differ from simple distillation?
-Fractional distillation is a more complex process than simple distillation, designed to separate components with boiling points that are close to each other, and it is generally more efficient and time-saving.
Outlines

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowMindmap

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowKeywords

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowHighlights

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowTranscripts

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowBrowse More Related Video
5.0 / 5 (0 votes)