Hybridization Theory (English)
Summary
TLDRThis script delves into the significance of hybridization theory in chemistry, essential for understanding molecular geometry and predicting chemical reactivity. It explains how the theory helps visualize molecules in 3D, using carbon as a basis to explore different hybridizations like sp3, sp2, and sp. The script covers the construction of molecules from 2D Lewis structures to 3D models, illustrating concepts with examples like methane, ethylene, and acetylene. It also touches on the impact of lone pairs and steric factors on bond angles, and the importance of hybridization in the broader field of chemistry.
Takeaways
- 📚 Hybridization theory was developed to explain the observed four bonds of carbon and to predict molecular geometry in three dimensions.
- 🧠 Understanding hybridization is essential for visualizing molecules in 3D, which is crucial for grasping chemical and physical properties and predicting reactivity.
- 📐 The theory introduces different hybridization states like sp3, sp2, and sp, which correspond to different bond angles and molecular geometries.
- 🌐 Hybridization allows chemists to understand the orientation of atoms within molecules, which is foundational to molecular geometry.
- 🔬 The carbon atom, with its four valence electrons, can form four covalent bonds through hybridization, contrary to the initial two unpaired electrons in the 2px and 2py orbitals.
- 📈 Hybrid orbitals are created by mixing atomic orbitals, resulting in new orbitals that are used for bonding and have specific spatial arrangements.
- 🛠 The sp3 hybridization results in a tetrahedral arrangement with bond angles of 109.5 degrees, as seen in methane (CH4) and ethane (C2H6).
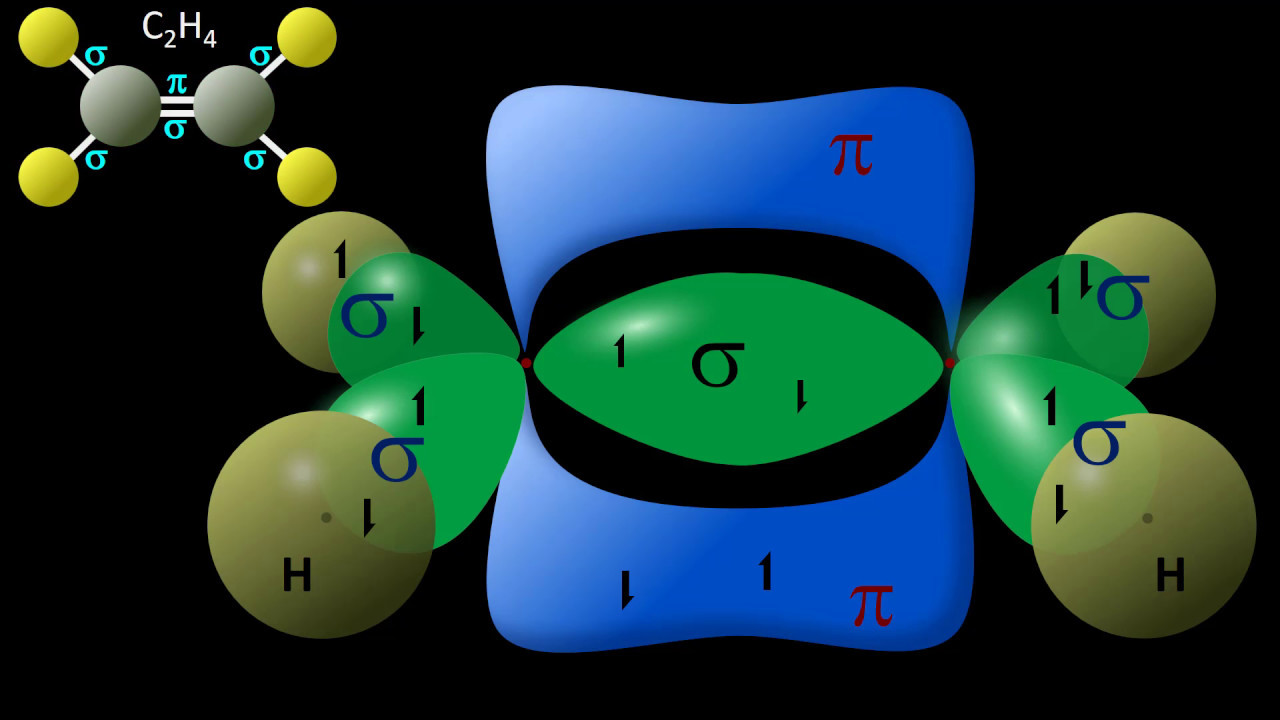
- 🔬 The sp2 hybridization leads to a trigonal planar arrangement with bond angles of 120 degrees, as found in ethylene (C2H4).
- 🌉 The sp hybridization results in a linear arrangement with bond angles of 180 degrees, as observed in acetylene (C2H2).
- 🔍 The concept of hybridization can be extended to other elements like nitrogen and oxygen, which also follow the principles of electron pair repulsion and orbital hybridization.
- 🔄 Hybridization theory also helps in understanding the existence of geometric isomers and the energy differences between them due to rotation restrictions around double and triple bonds.
Q & A
Why was hybridization theory developed?
-Hybridization theory was developed to better explain the observed four bonds of carbon and to predict overall molecular geometry, including bond angles in three dimensions.
Why is hybridization theory important in chemistry?
-Hybridization theory is important because it allows chemists to envision molecules in three dimensions, which is essential for understanding chemical properties, physical properties, and predicting chemical reactivity.
How does hybridization theory help in visualizing molecules?
-Hybridization theory provides a framework for understanding the orientation of atoms within molecules in three dimensions, which is crucial for predicting chemical behavior and reactivity.
What is the basic electron configuration of a carbon atom?
-A carbon atom has six electrons with the configuration 1s² 2s² 2p², where the valence or outermost electrons are responsible for bond making and breaking.
How does hybridization theory explain the bonding in methane (CH4)?
-In methane, the carbon atom is sp³ hybridized, which means it has four unpaired valence electrons capable of forming covalent bonds with four hydrogen atoms, resulting in a tetrahedral geometry with bond angles of 109.5 degrees.
What are the different types of hybrid orbitals a carbon atom can have?
-A carbon atom can have sp³, sp², and sp hybrid orbitals, each corresponding to different numbers of electron domains (four, three, and two, respectively) and different molecular geometries.
How does the shape of atomic orbitals influence hybridization?
-The shapes of atomic orbitals, such as the spherical 2s and dumbbell-shaped 2p orbitals, determine how they can mix to form hybrid orbitals, which in turn influences the three-dimensional shape of the molecule.
What is the significance of sigma and pi bonds in the context of hybridization?
-Sigma bonds are formed by the overlap of hybrid orbitals along an axis connecting two nuclei, while pi bonds result from the side-to-side overlap of unhybridized p orbitals. Understanding these bonds is crucial for predicting molecular geometry and reactivity.
How does the concept of hybridization apply to other elements besides carbon?
-The principles of hybridization discussed for carbon can also be applied to other elements, such as nitrogen and oxygen, by counting the number of groups (atoms or lone pairs) around the central atom to deduce the type of hybridization.
What is the role of VSEPR theory in understanding molecular geometry?
-Valence Shell Electron Pair Repulsion (VSEPR) theory allows chemists to predict deviations from ideal bond angles based on electron pair repulsions, including the effects of lone pairs and the presence of different groups around a central atom.
Can you provide an example of how hybridization theory helps in understanding the structure of a more complex molecule like tetrodotoxin?
-While the script does not provide specific details about tetrodotoxin, hybridization theory can be used to understand its complex molecular geometry by determining the hybridization states of its constituent atoms and predicting the orientation of its bonds and functional groups in three dimensions.
Outlines

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифMindmap

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифKeywords

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифHighlights

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифTranscripts

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифПосмотреть больше похожих видео
Hybrid Orbitals explained - Valence Bond Theory | Orbital Hybridization sp3 sp2 sp

MENENTUKAN BENTUK MOLEKUL BERDASARKAN TEORI DOMAIN ELEKTRON

Hibridisasi SP, SP2 dan SP3

VSEPR Theory and Molecular Geometry

Class 11 Chemistry | Chemical Bonding and Molecular Str. in 15 Minutes | Rapid Revision by BP Sir

An Introduction to Inorganic Chemistry- Lecture 2
5.0 / 5 (0 votes)
