6.5 Intermolecular Forces
Summary
TLDRThis video covers intermolecular forces, explaining how molecules interact through forces like dipole-dipole interactions, hydrogen bonding, and London dispersion forces. The strength of these forces affects properties like boiling points, with stronger forces leading to higher boiling points. Dipole-dipole forces occur between polarized molecules, while hydrogen bonds form between highly electronegative elements like oxygen, fluorine, or nitrogen and hydrogen. London dispersion forces, though weak and temporary, affect all atoms due to the random motion of electrons. The video excludes molecular geometry to focus on these key interactions.
Takeaways
- 🔬 Intermolecular forces refer to interactions between molecules, not the bonds within a single molecule.
- 🔥 Boiling points help measure the strength of intermolecular forces, with higher boiling points indicating stronger forces.
- 🏗️ Metallic compounds have the highest boiling points, followed by ionic compounds, and then molecular compounds.
- ⚡ Dipole-dipole forces occur between polarized molecules (dipoles), which have equal but opposite charges on either end.
- 🔁 Dipole-dipole forces are short-range and work only between adjacent molecules.
- 💧 Water is a strong dipole with a very negative oxygen atom and positive hydrogen atoms, while carbon dioxide is nonpolar due to balanced dipoles.
- 💥 Strong dipoles can induce slight polarity in nonpolar molecules, such as oxygen becoming soluble in water.
- 🌡️ Hydrogen bonds occur when hydrogen is attached to highly electronegative elements (fluorine, oxygen, nitrogen), leading to very polar substances.
- 🌀 London dispersion forces are weak, temporary forces that occur due to random electron movement in atoms.
- 🌬️ Noble gases like helium and neon rely on weak London dispersion forces, resulting in very low boiling points.
Q & A
What are intermolecular forces?
-Intermolecular forces measure interactions between molecules, not within the bonds of a molecule, but how one molecule affects another. They are responsible for holding molecules together in the liquid or solid state.
How do chemists measure the strength of intermolecular forces?
-Chemists measure the strength of intermolecular forces by boiling a substance. The amount of energy needed to convert a liquid into a gas indicates how strongly the molecules are attached to one another.
What is the relationship between boiling points and intermolecular forces?
-A higher boiling point indicates stronger intermolecular forces because more energy (in the form of heat) is needed to separate the molecules.
Which substances tend to have the highest boiling points?
-Metallic compounds tend to have the highest boiling points, followed by ionic compounds, and then molecular compounds.
What are dipole-dipole forces?
-Dipole-dipole forces occur between polarized molecules, called dipoles, which have equal but opposite charges separated by a short distance. These forces are short-range and work between adjacent molecules.
Why does iodine chloride have a higher boiling point than diatomic bromine?
-Iodine chloride has a higher boiling point because it is a polar compound with dipole-dipole forces, while diatomic bromine is nonpolar and lacks these forces, resulting in a lower boiling point.
How does the structure of water contribute to its polarity?
-Water is polar because the oxygen atom, being more electronegative, pulls the electrons away from the hydrogen atoms, creating a dipole with a negative charge on the oxygen side and positive charges on the hydrogen sides.
Why is carbon dioxide nonpolar despite having polar bonds?
-Although the bonds between carbon and oxygen in CO2 are polar, the dipoles are oriented in opposite directions and cancel each other out, resulting in a nonpolar molecule.
What are hydrogen bonds, and why are they strong?
-Hydrogen bonds occur when hydrogen is attached to highly electronegative elements like fluorine, oxygen, or nitrogen. The hydrogen essentially loses its electron, leaving a lone proton that is strongly attracted to the negative side of nearby dipoles, making the bond particularly strong.
What are London dispersion forces, and why are they weak?
-London dispersion forces are weak intermolecular forces that arise from the random movement of electrons, creating temporary dipoles. These forces are short-lived and weak, which is why substances relying on them, like noble gases, have low boiling points.
Outlines

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードMindmap

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードKeywords

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードHighlights

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードTranscripts

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレード関連動画をさらに表示

What are Intermolecular Forces?

Gaya antarmolekul (Kimia SMA/MA Kelas 10)

Intermolecular Forces | Chemistry

INTERMOLECULAR FORCES | Science 11 | Physical Science

Ikatan Kimia (9) | Gaya Antar Molekul | Ikatan Antar Molekul |Kimia kelas 10
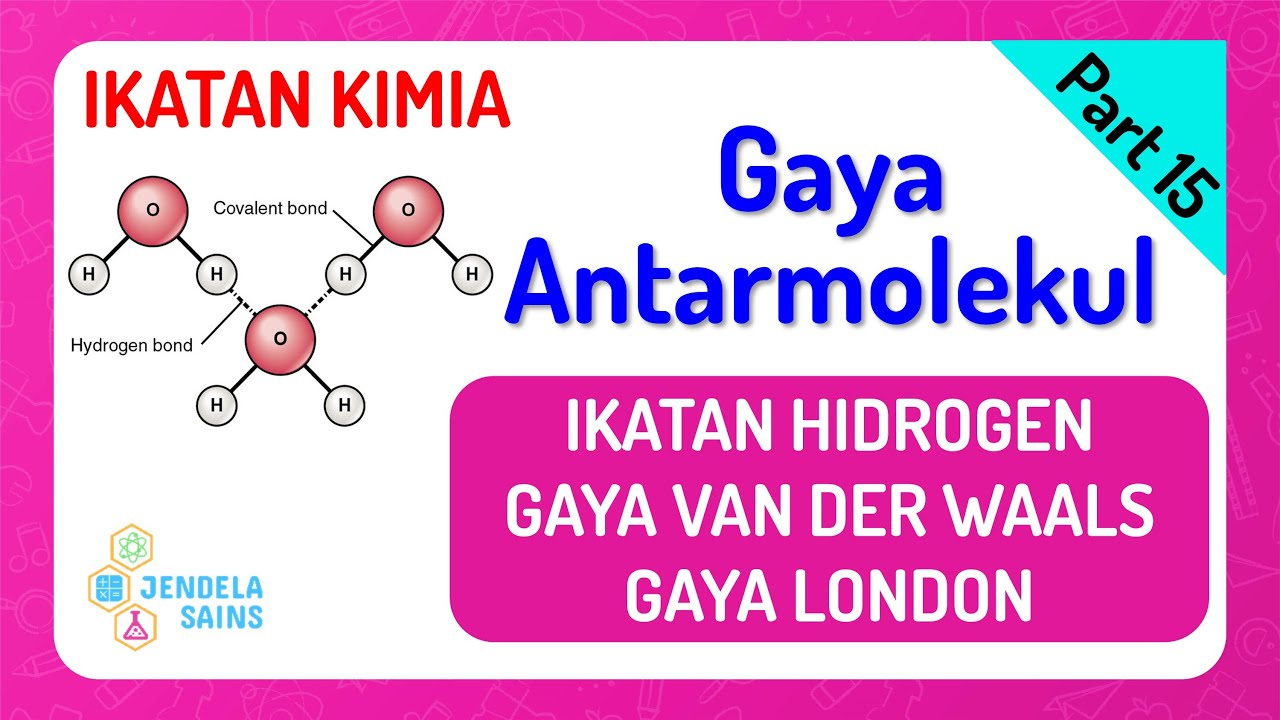
Ikatan Kimia • Part 15: Gaya Antar Molekul: Ikatan Hidrogen & van der Waals, Gaya London
5.0 / 5 (0 votes)
