Electron Configurations of Elements
Summary
TLDRThis video explains how to determine the electron configurations of lithium, sulfur, and cobalt by using the periodic table and block notation. It covers the basics of identifying elements in the s, p, and d blocks, using superscripts for electron counts, and how to use shorthand notation with noble gases. The tutorial walks through the process step-by-step, demonstrating how to navigate each row and block of the table, including the unique placement of the d-block elements. It's an easy-to-follow guide for understanding electron configurations and the periodic table's structure.
Takeaways
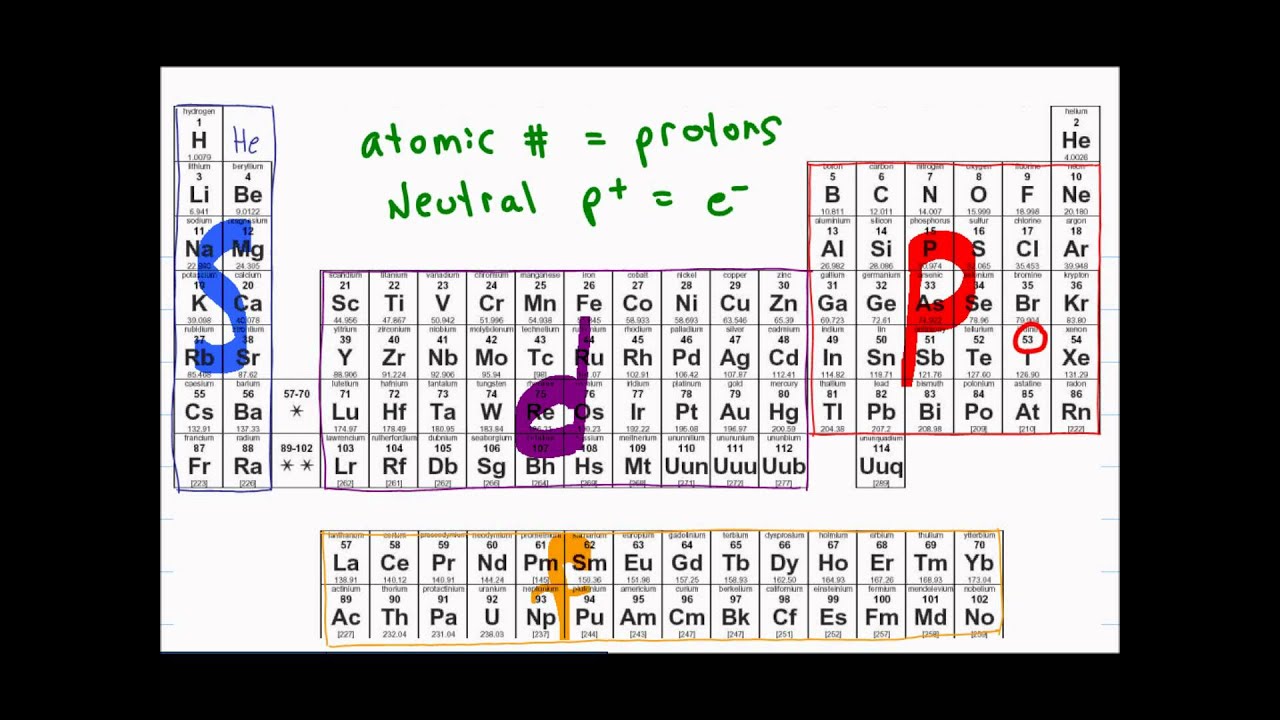
- 😀 The video explains how to find the electron configurations of elements using the periodic table.
- 😀 Elements are located on the periodic table, and their position determines the electron configuration.
- 😀 The periodic table is divided into blocks: s-block, p-block, and d-block, each with a different maximum number of electrons.
- 😀 s-block can hold up to 2 electrons, p-block up to 6, and d-block up to 10.
- 😀 To write electron configurations, you go row by row and assign electrons to the appropriate block.
- 😀 Lithium's electron configuration is 1s² 2s¹, showing the filling of the s-block.
- 😀 Sulfur's electron configuration is 1s² 2s² 2p⁶ 3s² 3p⁴, demonstrating filling in both s-block and p-block.
- 😀 For transition metals like Cobalt, shorthand notation using noble gases (e.g., [Ar]) simplifies electron configuration.
- 😀 Cobalt's electron configuration in shorthand is [Ar] 4s² 3d⁷, with valence electrons primarily in the outermost s-block.
- 😀 When removing electrons from d-block elements, electrons are removed first from the outermost s-block sublevel.
- 😀 Memorizing the block structure and row numbers of the periodic table is key to correctly writing electron configurations.
- 😀 The video emphasizes a step-by-step approach to electron configurations: locate element, determine block and row, assign electrons, and optionally use noble gas shorthand.
Q & A
What is the first step in determining the electron configuration of an element?
-The first step is to locate the element on the periodic table and identify its row and block (s, p, or d).
How do you know which sublevel (s, p, d) to start with when writing an electron configuration?
-The block of the element on the periodic table determines the sublevel: s-block → s sublevel, p-block → p sublevel, d-block → d sublevel.
How is the 1s sublevel filled for lithium?
-Lithium has two electrons in the 1s sublevel, written as 1s².
How many electrons can the s, p, and d blocks hold?
-The s-block can hold 2 electrons, the p-block can hold 6 electrons, and the d-block can hold 10 electrons.
What is the electron configuration of sulfur?
-Sulfur has the electron configuration 1s² 2s² 2p⁶ 3s² 3p⁴.
What is the purpose of using noble gas shorthand in electron configurations?
-Noble gas shorthand simplifies the electron configuration by representing all the electrons up to the previous noble gas in brackets, reducing the need to write the full sequence.
How do you write the shorthand electron configuration for cobalt?
-Cobalt can be written as [Ar] 4s² 3d⁷, using argon as the noble gas shorthand for the first 18 electrons.
Why do d-block elements often have their valence electrons in the s sublevel?
-Although d-block elements have electrons in the d sublevel, their outermost electrons are in the s sublevel of the highest principal energy level, so when forming ions, electrons are usually removed from the s sublevel first.
When filling the p-block for row 3 elements, how many electrons are placed in the 3p sublevel?
-The number of electrons in the 3p sublevel corresponds to the element's position in the p-block; for sulfur, which is the fourth element in the 3p block, there are 4 electrons in 3p.
What method is suggested for memorizing the blocks of the periodic table?
-The video suggests labeling each block (s, p, d) and memorizing their positions and electron capacities to easily write electron configurations.
Why does the 3d sublevel for cobalt start at 3d⁷ instead of including all previous electrons?
-Because cobalt’s first 18 electrons are represented by the noble gas argon in shorthand notation, the 3d sublevel only accounts for the remaining 7 electrons beyond argon.
How do you determine the superscript number for each sublevel in electron configuration?
-The superscript represents the number of electrons in that sublevel, based on counting from the element’s position in its block and row.
Outlines

Cette section est réservée aux utilisateurs payants. Améliorez votre compte pour accéder à cette section.
Améliorer maintenantMindmap

Cette section est réservée aux utilisateurs payants. Améliorez votre compte pour accéder à cette section.
Améliorer maintenantKeywords

Cette section est réservée aux utilisateurs payants. Améliorez votre compte pour accéder à cette section.
Améliorer maintenantHighlights

Cette section est réservée aux utilisateurs payants. Améliorez votre compte pour accéder à cette section.
Améliorer maintenantTranscripts

Cette section est réservée aux utilisateurs payants. Améliorez votre compte pour accéder à cette section.
Améliorer maintenantVoir Plus de Vidéos Connexes

Electron Configuration
Electron Configuration of Atoms + Shortcut Tutorial Video

A Level Chemistry Revision "Electron Configuration and the Periodic Table"

How to Write the Electron Configuration for an Element in Each Block

Família e período da Tabela Periódica através da Distribuição Eletrônica.

GCSE Chemistry Revision "Ionic Bonding 1: Ionic Bonding between Group 1 and Group 7"
5.0 / 5 (0 votes)
