GCSE Chemistry - What is Ionic Bonding? How Does Ionic Bonding Work? Ionic Bonds Explained #14
Summary
TLDRThis educational video delves into the formation of ionic bonds through the interaction of particles. It explains how ions, formed by atoms losing or gaining electrons to achieve stability, are depicted in equations and dot and cross diagrams. The video illustrates the transfer of electrons from a metal like sodium to a non-metal like chlorine, resulting in ions with full outer electron shells that attract each other due to opposite charges. The process is exemplified with the formation of magnesium chloride (MgCl2), highlighting the movement of electrons and the arrangement of ions in a compound. The video is an informative resource for understanding the basics of ionic bonding.
Takeaways
- 🔬 Ions are formed when atoms lose or gain electrons to achieve a stable electron configuration.
- ⚛️ Sodium loses one electron to become a sodium ion (Na⁺), while chlorine gains one electron to become a chloride ion (Cl⁻).
- 🧲 The transfer of electrons from a metal (like sodium) to a non-metal (like chlorine) results in ions with opposite charges that attract each other.
- 📚 Ionic bonds are strong electrostatic forces that hold together ions with opposite charges, similar in strength to covalent bonds.
- 📊 Dot and cross diagrams are used to represent the formation of ionic compounds, showing the transfer of electrons between atoms.
- 📝 In dot and cross diagrams, electrons from one atom are represented as dots, and from another as crosses, to distinguish their origin.
- 🎯 The movement of electrons is indicated by an arrow in the diagram, showing the direction of electron transfer.
- 🌟 It's important to show the full electron configuration of atoms in diagrams, but sometimes only the outermost shell needs to be depicted for simplicity.
- 🔋 Magnesium chloride (MgCl₂) is formed when magnesium donates two electrons to two chlorine atoms, resulting in a Mg²⁺ ion and two Cl⁻ ions.
- 📐 In diagrams with multiple ions, the ions are arranged to reflect their positions in the actual compound, showing their electrostatic attractions.
Q & A
What is an ion?
-An ion is an atom or molecule that has lost or gained electrons, resulting in a net electrical charge.
Why do atoms form ions?
-Atoms form ions to achieve a stable electron configuration, typically by having a full outer shell of electrons.
How does a sodium atom form an ion?
-A sodium atom forms a sodium ion by losing one electron, resulting in a sodium one plus ion (Na⁺).
What happens to a chlorine atom when it gains an electron?
-When a chlorine atom gains an electron, it forms a chloride ion with a one minus charge (Cl⁻).
What is the significance of having a full outer shell of electrons?
-Having a full outer shell of electrons is significant because it represents a stable electron configuration, which is energetically favorable for an atom.
What is an ionic bond?
-An ionic bond is a type of chemical bond formed by the electrostatic attraction between oppositely charged ions.
How are ionic bonds similar to covalent bonds?
-Ionic bonds are similar to covalent bonds in that they both involve the formation of a stable compound through the interaction of atoms, and they can be quite strong.
What is a dot and cross diagram?
-A dot and cross diagram is a way to represent the formation of ions and ionic compounds, showing the movement of electrons and the resulting charges on the ions.
Why are electrons represented differently in a dot and cross diagram?
-Electrons are represented differently (as dots and crosses) in a dot and cross diagram to distinguish which electrons belong to which atom, especially after the transfer of electrons.
Can you explain the steps to draw a dot and cross diagram for magnesium chloride (MgCl₂)?
-To draw a dot and cross diagram for MgCl₂, start by showing magnesium with two electrons in its outer shell and two chlorine atoms each needing one more electron for a full shell. Then, depict the transfer of electrons from magnesium to chlorine, resulting in a magnesium two plus ion (Mg²⁺) and two chloride one minus ions (2Cl⁻). Finally, arrange the ions to reflect their positions in the actual compound.
Why do electrons typically move from a metal to a non-metal during ionic bond formation?
-Electrons move from a metal to a non-metal during ionic bond formation because metals tend to lose electrons easily to achieve a stable configuration, while non-metals tend to gain electrons to complete their outer shells.
Outlines

Cette section est réservée aux utilisateurs payants. Améliorez votre compte pour accéder à cette section.
Améliorer maintenantMindmap

Cette section est réservée aux utilisateurs payants. Améliorez votre compte pour accéder à cette section.
Améliorer maintenantKeywords

Cette section est réservée aux utilisateurs payants. Améliorez votre compte pour accéder à cette section.
Améliorer maintenantHighlights

Cette section est réservée aux utilisateurs payants. Améliorez votre compte pour accéder à cette section.
Améliorer maintenantTranscripts

Cette section est réservée aux utilisateurs payants. Améliorez votre compte pour accéder à cette section.
Améliorer maintenantVoir Plus de Vidéos Connexes

Ikatan Kimia (2) | Ikatan Ion (Elektrovalen) | Kimia kelas 10

12.4 The Fundamental Types of Crystalline Solids

enlace covalente 1a parte
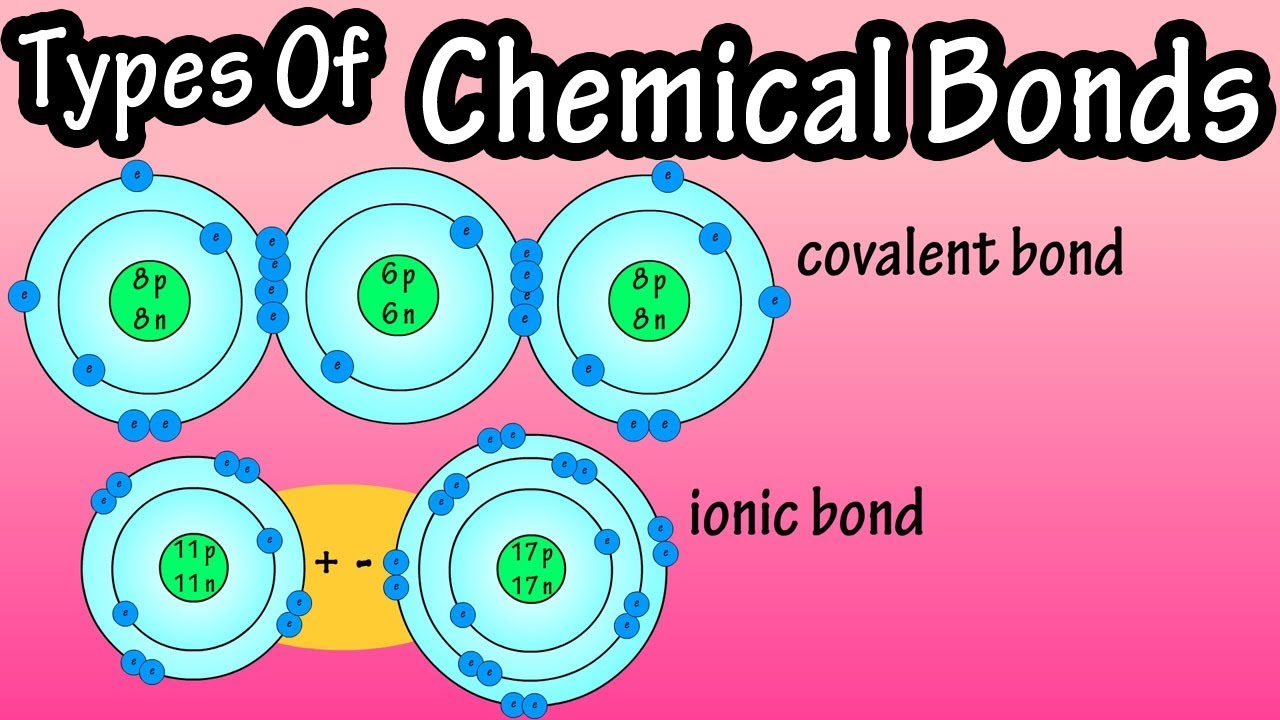
Types Of Chemical Bonds - What Are Chemical Bonds - Covalent Bonds And Ionic Bonds - What Are Ions

Legame Ionico - Legami Chimici | Lezioni di Chimica
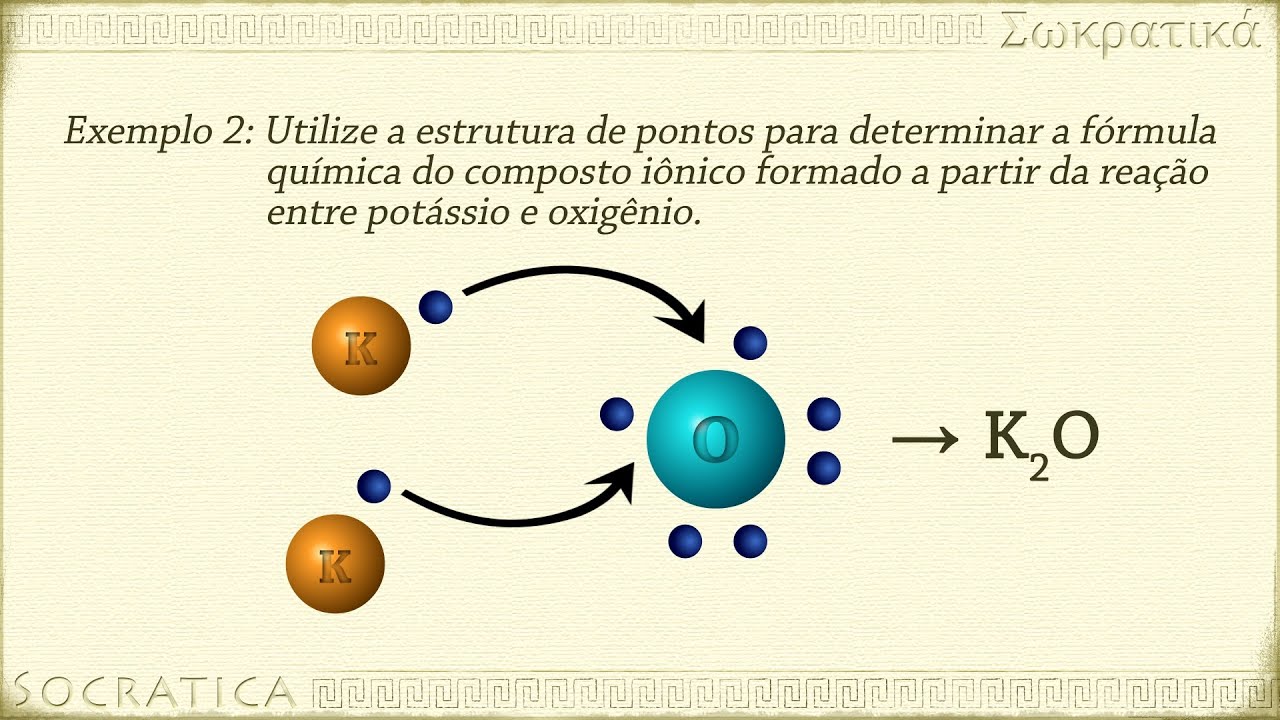
Química: Ligações Iônicas
5.0 / 5 (0 votes)
