FORÇA ELÉTRICA (LEI DE COULOMB) - Teoria e Exercícios
Summary
TLDRThis video explains the fundamental concepts of electric force, focusing on Coulomb's law, which describes the force between electric charges. The presenter explains attraction and repulsion between charges with opposite or like signs, the significance of the law’s constants, and the need for using magnitudes of charges. Through practical examples, including charge calculations and forces between charges, the video demonstrates the application of Coulomb’s law in real-world scenarios. The explanation helps viewers understand how to calculate the force and interpret problems involving electric forces in physics, with specific focus on typical exam problems.
Takeaways
- ⚡ Electric charges exert forces on each other, which can be either attractive or repulsive.
- ➕ Opposite charges attract each other, while like charges repel each other.
- 🧲 The forces between two charges are equal in magnitude and opposite in direction, following Newton's Third Law.
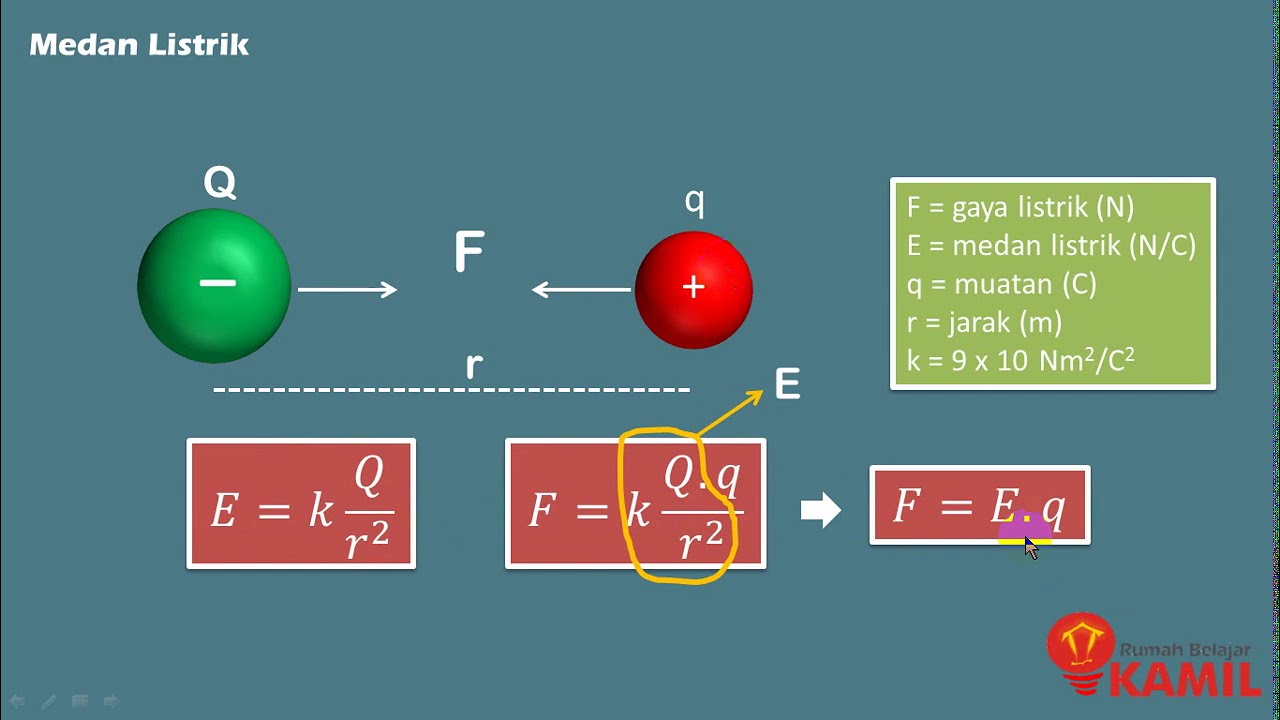
- 📏 Coulomb's Law calculates the magnitude of the electric force: F = k * |q1 * q2| / d².
- 🧮 The charges in Coulomb's Law are expressed in absolute value to ensure force is always positive.
- 🌐 The electrostatic constant k depends on the medium; in vacuum, k ≈ 9 × 10⁹ N·m²/C².
- 📐 Distance in Coulomb's Law must always be in meters, requiring conversion if given in centimeters or millimeters.
- 🟰 For a charge to remain in equilibrium between two others, the forces from both sides must be equal in magnitude and opposite in direction.
- ✖️ Many exam problems require analyzing situations and relationships between charges, not just direct substitution into formulas.
- 📊 When charges or distances change, the new force can be calculated using the ratio of previous known forces and distances.
- 💡 Understanding the concepts of attraction, repulsion, and force equilibrium is key to solving practical problems in electrostatics.
- 🔢 Simplifying algebraic expressions carefully, including square roots and squares, is essential for finding correct distances and forces.
Q & A
What happens between two charges with opposite signs?
-Two charges with opposite signs attract each other, creating a force that pulls them together.
What occurs when two charges have the same sign?
-Two charges with the same sign repel each other, meaning they push away from one another.
Why do we use the modulus (absolute value) of charges in Coulomb's law?
-The modulus is used to ignore the sign of the charges, ensuring the calculated force is always positive, since a negative force would not make physical sense.
What is Coulomb's law formula and what does each term represent?
-Coulomb's law is F = K * |q1| * |q2| / d^2, where F is the electric force, K is the electrostatic constant of the medium, |q1| and |q2| are the magnitudes of the charges, and d is the distance between the charges.
How does distance affect the magnitude of the electric force?
-The electric force decreases with the square of the distance between charges; doubling the distance reduces the force to one-fourth.
How do you determine the position of a charge in equilibrium between two other charges?
-A charge is in equilibrium when the forces acting on it from the other two charges are equal in magnitude and opposite in direction, which can be solved mathematically by equating the forces.
Why is it important to convert distances to meters when using Coulomb's law?
-The electrostatic constant K is defined using SI units, which requires distances in meters to ensure the force is correctly calculated in newtons.
In problem-solving, how can previously calculated forces help in new situations?
-Previously calculated forces can be used as known quantities to simplify calculations in proportional problems, allowing the use of ratios instead of recalculating everything from scratch.
Why should parentheses be carefully applied in the denominator of Coulomb's law?
-Parentheses are crucial because the distance term is squared; missing parentheses can lead to incorrect results by squaring only part of the expression instead of the entire distance.
What strategy can simplify solving the quadratic equations arising in equilibrium problems?
-If distances are positive, taking the square root of both sides can simplify the equation, eliminating squares and allowing easier calculation of distances.
What is the typical value of K in a vacuum and why is it often provided in exercises?
-K is approximately 9 × 10^9 N·m²/C² in a vacuum. Exercises provide it because its value depends on the medium, and students usually are not expected to memorize it.
Why is it rare for exercises to provide all values for direct substitution in Coulomb's law?
-Most exercises aim to test conceptual understanding and problem-solving skills, requiring students to interpret complex scenarios and use proportions or algebra rather than just plug in numbers.
Outlines

Esta sección está disponible solo para usuarios con suscripción. Por favor, mejora tu plan para acceder a esta parte.
Mejorar ahoraMindmap

Esta sección está disponible solo para usuarios con suscripción. Por favor, mejora tu plan para acceder a esta parte.
Mejorar ahoraKeywords

Esta sección está disponible solo para usuarios con suscripción. Por favor, mejora tu plan para acceder a esta parte.
Mejorar ahoraHighlights

Esta sección está disponible solo para usuarios con suscripción. Por favor, mejora tu plan para acceder a esta parte.
Mejorar ahoraTranscripts

Esta sección está disponible solo para usuarios con suscripción. Por favor, mejora tu plan para acceder a esta parte.
Mejorar ahoraVer Más Videos Relacionados
IPA Kelas 9 : Listrik Statis III (Medan Listrik)

11TH STD PHYSICS|10.ELECTROSTATICS|LECTURE 03|PRADEEP GIRI SIR

Listrik Statis, Konsep Hukum Coulomb dan Contoh-contoh Soal

Fisika kelas 12 | Listrik Statis (part 1) Muatan listrik dan gaya coulomb

Eduscribe : Fisika (Listrik Statis) Part 1

Fisika Dasar 2A - Gaya dan Medan Listrik
5.0 / 5 (0 votes)
