Konsentrasi Larutan (in Bahasa)
Summary
TLDRThis lesson covers various methods for determining the concentration of a solution, including molarity, molality, normality, dilution, PPM, and PPB. Key formulas and examples are provided to illustrate each method, such as calculating molarities from solid or liquid solutes, determining molality with a known solute and solvent weight, and understanding normality in acid-base and redox reactions. The lesson also explains how to dilute solutions and calculate concentrations in terms of PPM and PPB, making it a comprehensive guide for chemistry students.
Takeaways
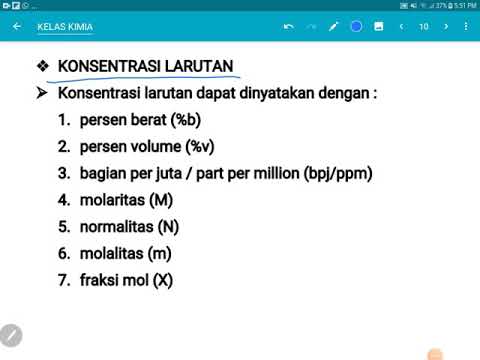
- 😀 Molarity (M) refers to the number of moles of solute per liter of solution, commonly used for concentrated solutions.
- 😀 The formula for molarity is M = moles of solute / volume of solution in liters, and it can be calculated using different methods depending on the state of the solute.
- 😀 Molality (m) measures the number of moles of solute per kilogram of solvent, and is important for temperature-independent concentration calculations.
- 😀 Normality (N) is used to measure the equivalent of solute in a solution, particularly for acid-base or redox reactions, and is calculated as Normality = Molarity × Equivalents.
- 😀 Dilution can be calculated using the formula C₁V₁ = C₂V₂, where C is concentration and V is volume. This formula helps adjust the concentration of a solution.
- 😀 PPM (Parts Per Million) is used to describe concentrations of solutes in large volumes of solvent, typically for concentrations in water, and calculated as PPM = (mg of solute) / (liters of solution).
- 😀 PPB (Parts Per Billion) is similar to PPM, but for much lower concentrations, where solute is measured in micrograms per liter of solution.
- 😀 Molarity, molality, and normality are all essential for determining the concentration of solutions, each suited for different types of chemical reactions.
- 😀 The script covers multiple example problems to apply the formulas for calculating molarity, normality, dilution, and PPM/PPB in real-life scenarios.
- 😀 Understanding the differences between molarity and molality is crucial for applying the right method depending on whether the concentration is based on volume (molarity) or mass (molality).
Q & A
What is molarity, and how is it calculated?
-Molarity (M) is the number of moles of a solute dissolved in one liter of solution. It is calculated using the formula: M = (moles of solute) / (volume of solution in liters).
How do you determine the molar mass (Mr) of a compound like CaCO3?
-To calculate the molar mass of CaCO3, you sum the atomic masses of its components: Ca (40 g/mol), C (12 g/mol), and O (16 g/mol, multiplied by 3 for three oxygen atoms). So, Mr of CaCO3 = 40 + 12 + (16 * 3) = 100 g/mol.
What is the difference between molarity and molality?
-Molarity (M) measures the concentration of a solute in terms of moles per liter of solution, while molality (m) measures the concentration of a solute in terms of moles per kilogram of solvent. Molarity depends on solution volume, while molality depends on the mass of the solvent.
What is the formula for molality?
-The formula for molality is: Molality (m) = (mass of solute in grams) / (molar mass of solute in g/mol) * (mass of solvent in kilograms).
How is normality (N) different from molarity?
-Normality (N) is a measure of concentration based on the equivalent of a solute, such as the number of hydrogen ions (H+) in an acid. It is calculated as N = Molarity * Equivalent factor. For example, sulfuric acid (H2SO4) has an equivalent factor of 2 because it can donate two H+ ions.
How do you calculate normality for an acid like H2SO4?
-To calculate the normality of an acid like H2SO4, multiply its molarity by its equivalent factor. Since H2SO4 donates two H+ ions, its equivalent factor is 2. For example, if the molarity is 0.25 M, the normality would be 0.25 M * 2 = 0.5 N.
What is the relationship between molarity and normality?
-The relationship between molarity (M) and normality (N) is given by: N = M * equivalent factor. The equivalent factor depends on the type of reaction (e.g., acid-base or redox) and how many ions are involved in the reaction.
What is the purpose of dilution, and how is it calculated?
-Dilution is the process of reducing the concentration of a solution by adding solvent. It is calculated using the formula: C1 * V1 = C2 * V2, where C1 and V1 are the initial concentration and volume, and C2 and V2 are the final concentration and volume.
How do you calculate the final concentration after diluting a solution?
-To calculate the final concentration after dilution, use the dilution equation: C1 * V1 = C2 * V2. For example, if you dilute 10 mL of 2 N HCl with 100 mL of water, the final concentration (C2) is 0.18 N.
What is the meaning of PPM and how is it calculated?
-PPM (Parts Per Million) is a measure of the concentration of a solute in a solution. It is calculated as: PPM = (mass of solute in milligrams) / (volume of solution in liters). For example, if 2,500 mg of NaCl is dissolved in 10 liters of water, the concentration is 250 PPM.
Outlines

此内容仅限付费用户访问。 请升级后访问。
立即升级Mindmap

此内容仅限付费用户访问。 请升级后访问。
立即升级Keywords

此内容仅限付费用户访问。 请升级后访问。
立即升级Highlights

此内容仅限付费用户访问。 请升级后访问。
立即升级Transcripts

此内容仅限付费用户访问。 请升级后访问。
立即升级浏览更多相关视频
Kelas Kimia : Konsentrasi Larutan (% berat, % volume, ppm / bpj)

MOLARIDAD (M), molalidad (m) y partes por millón (ppm)

Kimia Analitik - Pendahuluan

Aula 7 - Concentração das soluções (Partes por milhão, ppm e Partes por bilhão, ppb)

Difference between Molarity and Molality

Ch 4 part 4 4 5 concentration of solutions
5.0 / 5 (0 votes)
