Gugus Fungsi senyawa karbon/senyawa organik - Kimia SMA kelas 12
Summary
TLDRIn this video, we explore the concept of functional groups in organic compounds, focusing on seven key types: haloalkanes, alcohols, ethers, aldehydes, ketones, carboxylic acids, and esters. Each functional group is discussed in terms of its molecular structure, IUPAC naming conventions, and common uses, such as alcohols for antiseptics and carboxylic acids for preservatives. The video also introduces the concept of functional isomers, where compounds share the same molecular formula but differ in their functional groups, such as alcohol vs ether and aldehyde vs ketone. A great primer for understanding organic chemistry!
Takeaways
- 😀 Functional groups are specific atoms or collections of atoms in organic compounds that determine their chemical properties.
- 😀 Alcohol is commonly associated with intoxicating drinks, but it also has various uses like antiseptic liquids and industrial solvents.
- 😀 Formalin, a preserved animal specimen, is an example of alcohol's functional group in practice.
- 😀 Functional groups are the most reactive parts of a carbon compound, which is why they determine the compound's behavior.
- 😀 There are multiple types of functional groups, each with a unique structure and chemical properties.
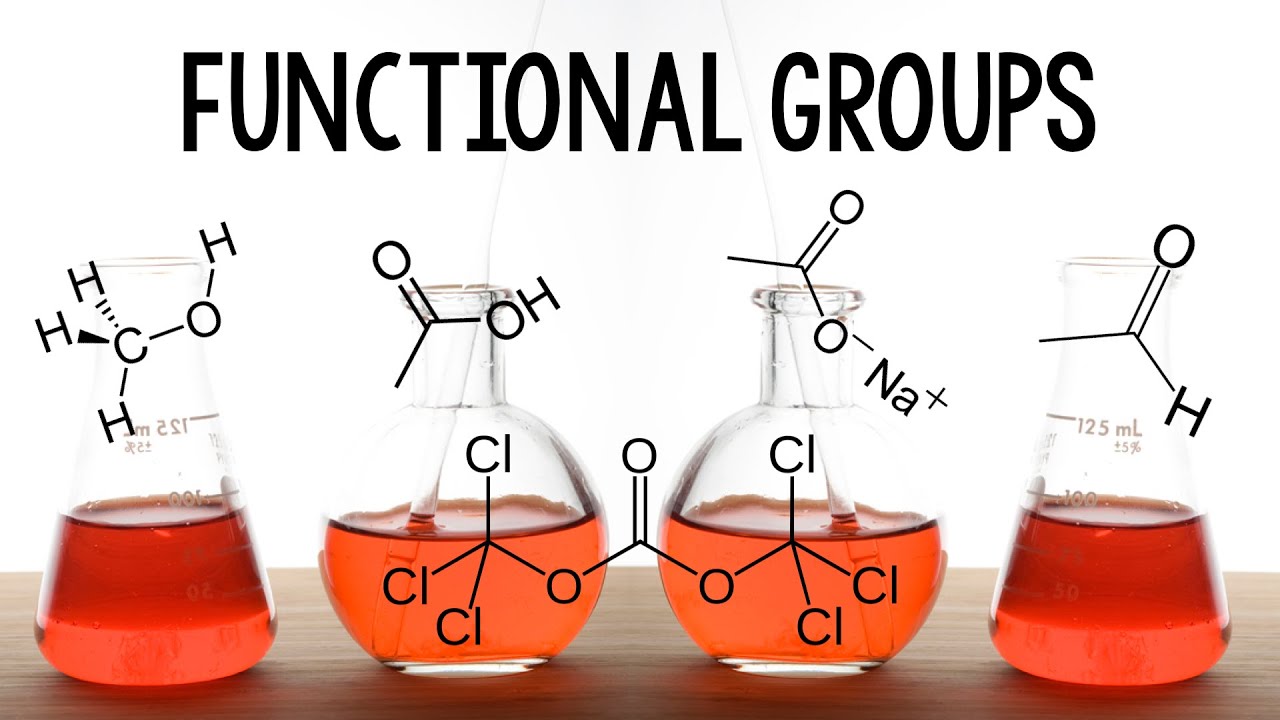
- 😀 A haloalkane (X) contains a halogen (F, Cl, Br, I) and is typically classified as an alkyl halide.
- 😀 Alkanols, or alcohols, have a hydroxyl group (-OH) and are classified as alcohols in IUPAC nomenclature.
- 😀 Ethers (R-O-R') have an oxygen atom connected to two alkyl groups and are known as alkoxy alkanes.
- 😀 Aldehydes (R-CHO) and ketones (R-CO-R') both contain a carbonyl group but differ in structure and functional group placement.
- 😀 Carboxylic acids (R-COOH) are known for their -COOH functional group and are common in substances like aspirin and benzoic acid.
- 😀 Esters (R-COOR) are characterized by a -COOR functional group and are often used in food flavorings and fragrances.
- 😀 Functional isomers are organic compounds that share the same molecular formula but differ in their functional groups, such as alcohols vs. ethers, aldehydes vs. ketones, and carboxylic acids vs. esters.
Q & A
What is a functional group in organic chemistry?
-A functional group is an atom or a collection of atoms within a molecule that determines the chemical properties and reactivity of that molecule. It is the most reactive part of a carbon compound.
How do functional groups influence organic compounds?
-Functional groups determine the nature of organic compounds by dictating their chemical behavior, reactivity, and interactions with other molecules.
What are haloalkanes, and how are they identified?
-Haloalkanes are organic compounds containing a halogen (F, Cl, Br, I) attached to an alkyl group. They are identified by the functional group RX, where R is an alkyl group and X is a halogen.
What is the IUPAC name for alcohols, and what is its functional group?
-The IUPAC name for alcohols is 'alkanol.' The functional group in alcohols is the hydroxyl group (-OH), which is attached to an alkyl group.
How does the structure of ethers differ from alcohols?
-Ethers have an oxygen atom bonded to two alkyl groups (R-O-R'). Alcohols, on the other hand, have a hydroxyl group (-OH) attached to an alkyl group.
What are the differences between aldehydes and ketones?
-Aldehydes contain a carbonyl group (C=O) attached to a hydrogen atom (R-CHO), whereas ketones have the carbonyl group bonded to two carbon atoms (R-CO-R').
What is the general formula for carboxylic acids, and what is their functional group?
-The general formula for carboxylic acids is R-COOH. Their functional group is the carboxyl group (-COOH), which includes both a carbonyl group (C=O) and a hydroxyl group (-OH).
What are esters, and how are they formed?
-Esters are organic compounds formed by the reaction between an alcohol and a carboxylic acid. Their general formula is R-COOR', where R and R' are alkyl groups.
Can two different functional groups share the same molecular formula?
-Yes, two different functional groups can have the same molecular formula but different structures. These are called functional isomers. For example, alcohols and ethers both have the formula CnH2n+2O but differ in their functional groups.
What is the relationship between carboxylic acids and esters in terms of functional isomerism?
-Carboxylic acids and esters both share the same molecular formula, CnH2nO2, but differ in their functional groups. Carboxylic acids have a carboxyl group (-COOH), while esters have an ester group (-COOR').
Outlines

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowMindmap

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowKeywords

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowHighlights

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowTranscripts

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowBrowse More Related Video

Tata Nama Senyawa Turunan Alkana | KIMIA KELAS 12

Hydrocarbon Derivatives

Mengenal GUGUS FUNGSI SENYAWA TURUNAN ALKANA, mulai dari Haloalkana sampai ESTER ‼️
Intro to Functional Groups

Senyawa Karbon Turunan Alkana • Part 1: Gugus Fungsi, Struktur, dan Deret Homolog

Funções orgânicas oxigenadas - Brasil Escola
5.0 / 5 (0 votes)