Mass Spectrometry MS
Summary
TLDRThis video explains how mass spectrometry allows chemists to measure the mass of atoms and molecules by creating ions and analyzing their deflection in electric and magnetic fields. It covers the key steps: sample introduction, ionization, acceleration, deflection, and detection, using limonene as an example. Fragmentation patterns reveal molecular structure, while high-resolution spectra can distinguish molecules with nearly identical masses. The video also highlights practical applications, including coupling mass spectrometry with gas chromatography (GC-MS) to analyze complex mixtures, such as natural extracts, demonstrating how major components and impurities can be identified with precision and minimal sample quantities.
Takeaways
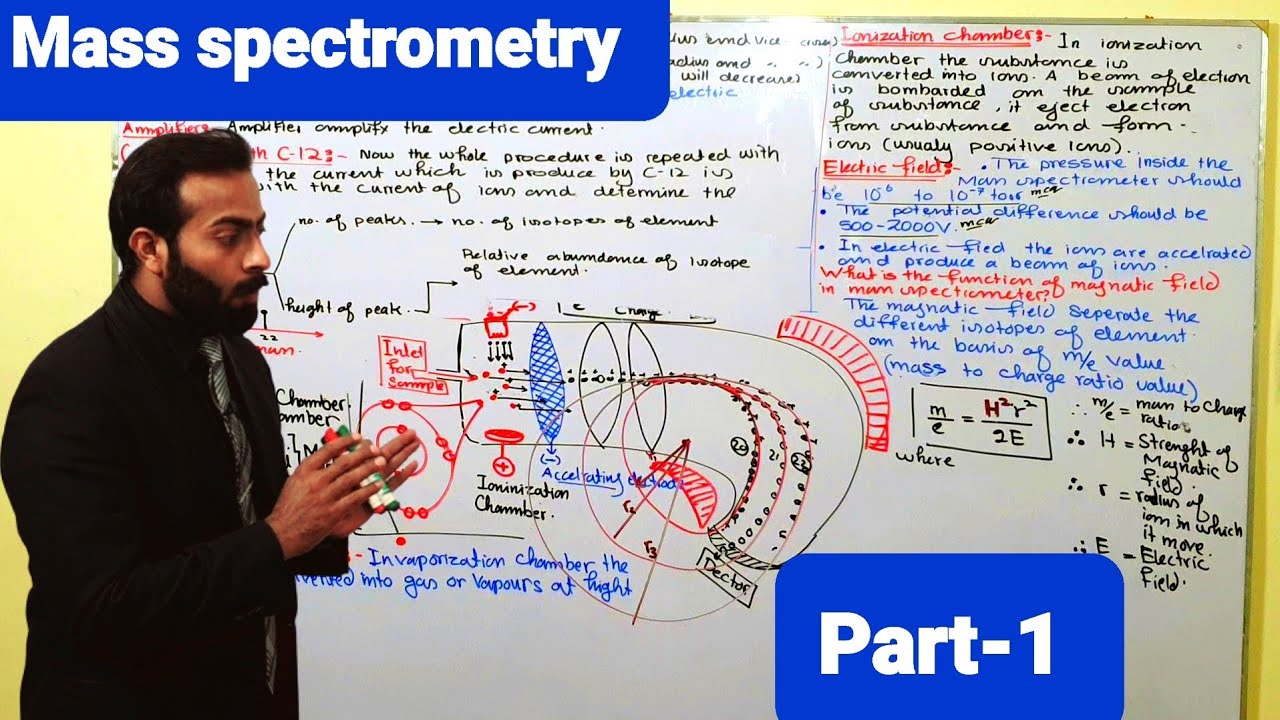
- ⚡ Mass spectrometry measures the mass-to-charge ratio of ions, not by directly weighing molecules.
- 🧪 All mass spectrometers operate on the principle of ion formation, acceleration, and deflection.
- 🔬 Magnetic sector instruments use combinations of electric and magnetic fields to separate ions based on mass.
- 💥 Ions are generated by bombarding sample molecules with high-energy electrons, creating positive ions.
- 🛤️ Accelerated ions are deflected through curved electric plates and magnetic fields to reach the detector.
- 📊 The mass spectrum displays peaks representing ions; peak height reflects relative abundance.
- 🧩 Fragment ions provide clues about the molecular structure, while the parent ion represents the intact molecule.
- 🎯 High-resolution mass spectrometry can differentiate molecules with the same nominal mass using precise atomic mass measurements.
- 🌿 Limonene, a natural compound from citrus peels, can be analyzed with very small sample amounts using mass spectrometry.
- 🔗 Gas chromatography-mass spectrometry (GC-MS) couples separation and detection, allowing identification of components in mixtures.
- 💨 Volatile samples vaporize under vacuum, while less volatile samples can be heated to produce ions.
- 🖥️ Mass spectra can be displayed to the nearest whole number or with three decimal places for higher precision.
Q & A
What is the basic principle behind how a mass spectrometer works?
-Mass spectrometers work by measuring the acceleration and deflection of ions formed from atoms and molecules in electric and magnetic fields. The ions' behavior in these fields helps determine their mass-to-charge ratio, which is used to identify the molecules.
How does the magnetic sector mass spectrometer operate?
-In a magnetic sector mass spectrometer, ions are first generated by bombarding the sample with a beam of electrons. These ions are then accelerated by electric fields, deflected by both electric and magnetic fields, and detected based on their mass-to-charge ratios.
Why is it important to maintain a high vacuum in the mass spectrometer?
-A high vacuum is crucial to prevent air molecules from colliding with the ions, allowing them to move at very high speeds (up to several kilometers per second) without interference, ensuring accurate measurements.
What does the mass spectrum of a molecule represent?
-A mass spectrum displays peaks that represent ions with specific masses. The peaks are plotted with mass-to-charge ratios on the horizontal axis and ion abundance on the vertical axis, which helps in identifying the molecular structure.
What is the role of fragmentation in mass spectrometry?
-Fragmentation occurs when the ions break apart into smaller charged fragments. The pattern of these fragments can provide valuable clues about the structure of the original molecule.
How does the mass spectrometer detect ions of different masses?
-By varying the strength of the electric and magnetic fields, the mass spectrometer can make ions of different masses reach the detector at different times, allowing for the identification of their mass-to-charge ratios.
What is the significance of the molecular ion in a mass spectrum?
-The molecular ion represents the intact molecule that has lost an electron during ionization. It is typically the ion with the largest mass in the spectrum, and it provides information about the molecular weight of the compound.
How are volatile and less volatile samples handled in mass spectrometry?
-Volatile samples can be directly vaporized inside the instrument, while less volatile samples are dissolved in a solvent (like dichloromethane) and then vaporized by heating within the instrument.
What is a high-resolution mass spectrum, and how is it different from a simple mass spectrum?
-A high-resolution mass spectrum provides measurements to three decimal places, allowing for precise differentiation between molecular formulas that have the same mass to the nearest whole number, such as distinguishing C10H16 from C9H12O.
What is the purpose of using gas chromatography in conjunction with mass spectrometry (GCMS)?
-Gas chromatography is used to separate different components of a sample based on their volatility. The separated components are then fed directly into the mass spectrometer for mass analysis, making GCMS a powerful technique for analyzing complex mixtures.
Outlines

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowMindmap

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowKeywords

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowHighlights

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowTranscripts

This section is available to paid users only. Please upgrade to access this part.
Upgrade Now5.0 / 5 (0 votes)