Periodic Table #2. Group 1, Alkali Metals
Summary
TLDRThis video explains the properties and reactivity of Group 1 elements in the periodic table, also known as alkali metals. It explores their tendency to lose a single valence electron, making them highly reactive, particularly with water. The script introduces the octet rule, showing how elements strive for a stable electron configuration, and highlights the characteristics of alkali metals like their low density, softness, and ability to form strong bases. The video also provides a memorable way to remember the alkali metals, from Hydrogen to Francium, and sets the stage for further exploration of these metals in future episodes.
Takeaways
- 😀 The periodic table can be imagined as a building with two towers: a thin one on the left and a wider one on the right, connected by the block of transition metals.
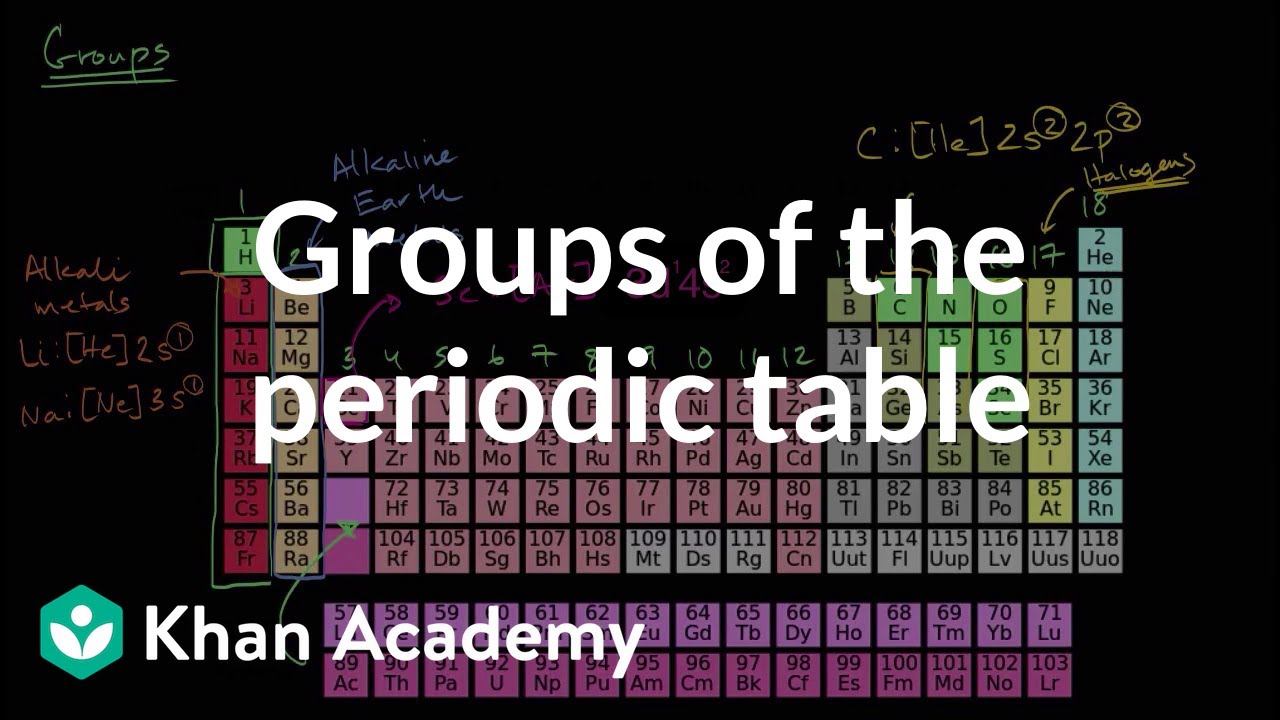
- 😀 Elements are arranged in rows (periods) and columns (groups) on the periodic table, with elements in the same group having similar properties.
- 😀 Elements in the same group have the same number of valence electrons, which are responsible for their chemical properties.
- 😀 The octet rule states that atoms are most stable when they have 8 electrons in their valence shell, similar to having a full bill of eight coins.
- 😀 Group 1 elements, which have one valence electron, are highly reactive and tend to lose that electron easily.
- 😀 Elements in Group 1 are called alkali metals, and they form strong bases like sodium hydroxide and potassium hydroxide when reacting with water.
- 😀 Alkali metals are soft, shiny, and have low density. Lithium, sodium, and potassium are less dense than water, with lithium being the lightest metal.
- 😀 Alkali metals react violently with water, producing hydroxides and hydrogen gas, making them impossible to find in nature in their pure form.
- 😀 As you move down Group 1 in the periodic table, the valence electron is located in higher energy levels, making these elements more reactive.
- 😀 The video introduces the first elements in Group 1: hydrogen, lithium, sodium, and potassium, followed by rubidium, cesium, and francium.
- 😀 The presenter plans to demonstrate how lithium reacts with water in the next episode, showing how alkali metals interact with water.
Q & A
What is the structure of the periodic table as described in the script?
-The periodic table is visualized as a building with two towers: a thin one on the left and a wider one on the right. Both towers are connected by the block of transition metals.
What are the two key organizing principles of the periodic table?
-The periodic table is organized into rows, known as periods, and columns, known as groups. Elements in the same group share similar properties, especially the number of valence electrons.
What are valence electrons and why are they important?
-Valence electrons are the electrons in the outermost shell of an atom. They are important because they determine an element's chemical properties, including how it reacts with other substances.
What is the octet rule, and how does it relate to the reactivity of elements?
-The octet rule states that atoms are most stable when they have eight electrons in their outermost shell. Elements tend to lose or gain electrons to achieve this stable configuration. This is especially relevant to alkali metals, which easily lose their single valence electron.
What are alkali metals, and why are they so reactive?
-Alkali metals are elements in Group 1 of the periodic table. They are highly reactive because they have one electron in their outermost shell, which they easily lose, making them prone to forming positive ions.
What is the significance of hydrogen in the context of alkali metals?
-Although hydrogen is in Group 1, it behaves differently from the alkali metals. It is often considered separately and is described as an 'antenna' on top of the periodic table building.
How do alkali metals react with water, and what do they form as a result?
-Alkali metals react violently with water to form hydroxides and hydrogen gas. For example, sodium reacts with water to form sodium hydroxide (NaOH) and hydrogen gas (H2).
What makes alkali metals less abundant in nature?
-Alkali metals are not found in their pure form in nature because they are highly reactive. Instead, they are usually found combined with other elements, such as in hydroxides, chlorides, and carbonates.
How does the reactivity of alkali metals change as you move down the group?
-As you move down Group 1, the elements have more electron shells, making the outermost electron farther from the nucleus and easier to lose. This increases their reactivity. For example, cesium is more reactive than lithium.
What mnemonic can help remember the alkali metals in order?
-A helpful mnemonic to remember the alkali metals in order is: 'Hydrogen, Lithium, Sodium, Potassium, Rubidium, Cesium, Francium.' This phrase makes it easier to recall the sequence of elements.
Outlines

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowMindmap

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowKeywords

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowHighlights

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowTranscripts

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowBrowse More Related Video

GCSE Chemistry - Modern Periodic Table #9

SPM Chemistry Form 4 (Periodic Table Of Elements) Chapter 4 - Part 2 Complete Revision
Groups of the periodic table | Periodic table | Chemistry | Khan Academy

5.2 Electron Configuration and the Periodic Table

The Periodic Table Song (2018 Update!) | SCIENCE SONGS

Grupo de metales alcalinos en la tabla periódica
5.0 / 5 (0 votes)