Sigma and Pi Bonds Explained, Basic Introduction, Chemistry
Summary
TLDRThis video script offers an educational introduction to sigma and pi bonds, essential concepts in chemistry. It explains that single bonds are sigma bonds, while double and triple bonds contain one and two pi bonds, respectively, in addition to sigma bonds. Using ethene as an example, the script illustrates how sigma bonds form from the overlap of atomic orbitals and how pi bonds arise from unhybridized p orbitals. The tutorial proceeds with practice problems involving molecules like acetylene, formaldehyde, and SO2Cl2, guiding viewers to determine the number of sigma and pi bonds in each compound.
Takeaways
- 🧬 Sigma and pi bonds are fundamental in understanding molecular structures, with sigma bonds being the first bond formed between atoms.
- 🔗 A single bond is always a sigma bond, while double and triple bonds contain additional pi bonds.
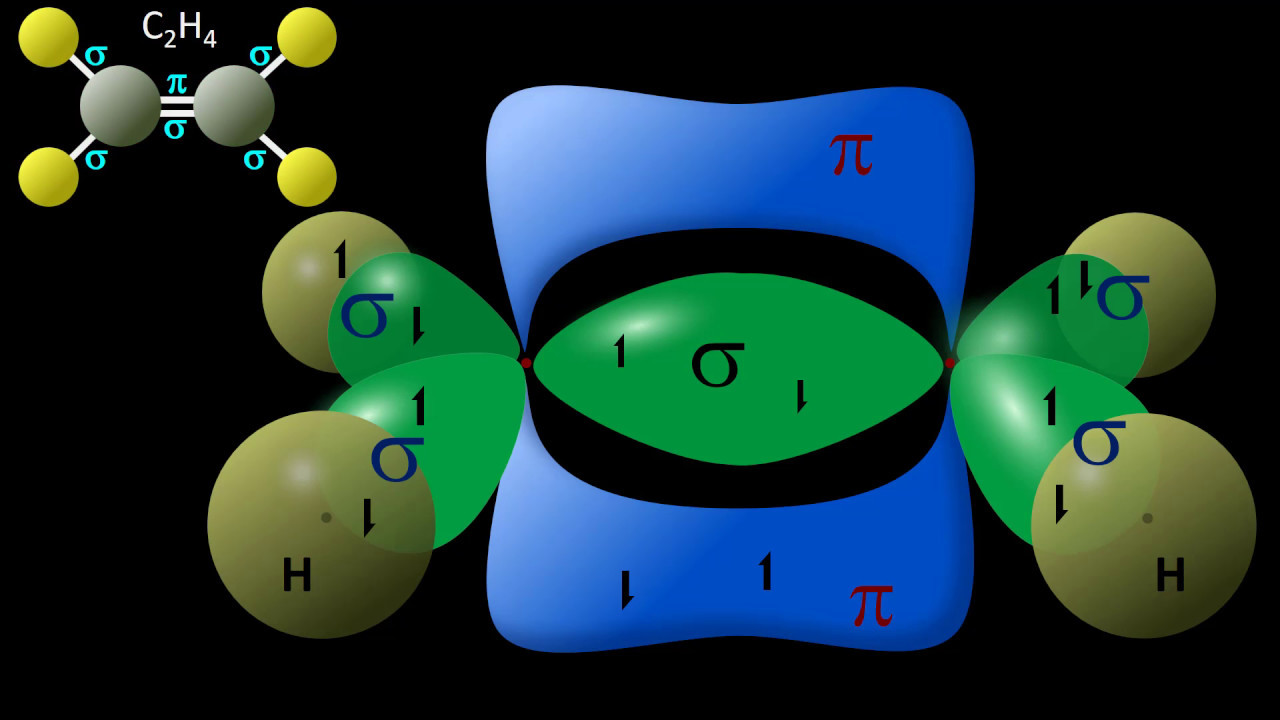
- 🌐 Ethene serves as an example where its Lewis structure shows sigma bonds formed from the overlap of atomic orbitals, with carbon being sp2 hybridized and hydrogen having s hybridization.
- 📊 The molecule ethene has five sigma bonds and one pi bond, which arises from the unhybridized p orbitals.
- 📚 The video script encourages viewers to practice by drawing the Lewis structure of acetylene (C2H2) and identifying its sigma and pi bonds.
- ✏️ Acetylene's Lewis structure reveals it to be a linear molecule with three sigma bonds and two pi bonds due to its triple bond.
- 📝 The script provides a step-by-step approach to determining the number of sigma and pi bonds in a molecule, starting with the Lewis structure.
- 💡 For molecules like formaldehyde (CH2O), the Lewis structure shows three sigma bonds and one pi bond within the double bond.
- 🧩 In SO2Cl2, the Lewis structure illustrates four single sigma bonds and two pi bonds from the double bonds, totaling six sigma and two pi bonds.
- 🔢 The script concludes with a more complex molecule, emphasizing the method of counting bonds to determine the number of sigma and pi bonds, resulting in 11 sigma and three pi bonds.
- 📘 The importance of understanding the hybridization of atoms and the types of bonds they form is highlighted for correctly identifying sigma and pi bonds in various molecules.
Q & A
What is a sigma bond?
-A sigma bond is a single bond formed from the overlap of atomic orbitals, typically resulting from the overlap of sp2 hybrid orbitals in carbon and s orbitals in hydrogen.
How many sigma and pi bonds are present in a double bond?
-A double bond contains one sigma bond and one pi bond.
What is the structure of ethene in terms of sigma and pi bonds?
-Ethene has five sigma bonds and one pi bond. The sigma bonds are formed from the overlap of sp2 hybrid orbitals and s orbitals, while the pi bond arises from the overlap of unhybridized p orbitals.
How do sigma and pi bonds differ in terms of orbital overlap?
-Sigma bonds result from the end-to-end overlap of orbitals, while pi bonds form from the side-to-side overlap of unhybridized p orbitals above and below the bond axis.
What is the Lewis structure of acetylene, and how many sigma and pi bonds does it contain?
-The Lewis structure of acetylene (C2H2) shows a linear molecule with one triple bond between the carbon atoms and two single bonds with hydrogen. It contains three sigma bonds and two pi bonds.
What is the number of sigma and pi bonds in formaldehyde (CH2O)?
-Formaldehyde (CH2O) has three sigma bonds and one pi bond.
How do you determine the number of sigma and pi bonds in a molecule?
-To determine the number of sigma and pi bonds, count all the single bonds as sigma bonds. Each double bond has one sigma and one pi bond, and each triple bond has one sigma and two pi bonds.
What is the Lewis structure of SO2Cl2, and how many sigma and pi bonds does it have?
-The Lewis structure of SO2Cl2 (sulfuryl chloride) has four sigma bonds and two pi bonds. The sigma bonds are formed between sulfur, oxygen, and chlorine atoms, while the double bonds contribute to the pi bonds.
Why is it important to know the number of sigma and pi bonds in a molecule?
-Knowing the number of sigma and pi bonds in a molecule helps in understanding its structural properties, bond strength, and reactivity.
How many sigma and pi bonds are present in a molecule with 11 sigma bonds and a triple and double bond?
-In such a molecule, there are 11 sigma bonds and three pi bonds (two from the triple bond and one from the double bond).
Outlines

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифMindmap

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифKeywords

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифHighlights

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифTranscripts

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифПосмотреть больше похожих видео

1. Introdução a Química Orgânica: Histórico e o Carbono (1/4) [Química Orgânica]

BTEC Applied Science - Unit 5 Chemistry - sigma and pi bonding hybridisation

Sigma and Pi Bonds (A-Level Chemistry)

Hybridization
Hybrid Orbitals explained - Valence Bond Theory | Orbital Hybridization sp3 sp2 sp

Kurikulum Merdeka Rangkuman Materi IPA Kelas 8 Bab 5 Unsur Senyawa dan Campuran
5.0 / 5 (0 votes)
