What are Molecules?
Summary
TLDRThis video script delves into the concept of molecules, explaining that they are groups of two or more atoms held together by chemical bonds. It clarifies that molecules can be homonuclear or heteronuclear, with water as an example of the latter. The script emphasizes molecules' neutrality and varying sizes, noting that while most are microscopic, some polymers like DNA can be macroscopic. It further explores the formation of molecules through covalent and ionic bonding, driven by electron sharing or transfer, and underscores the importance of quantum mechanics in understanding these bonds. The script concludes by framing molecules as a scientific observation tool for atomic interactions rather than a fundamental entity.
Takeaways
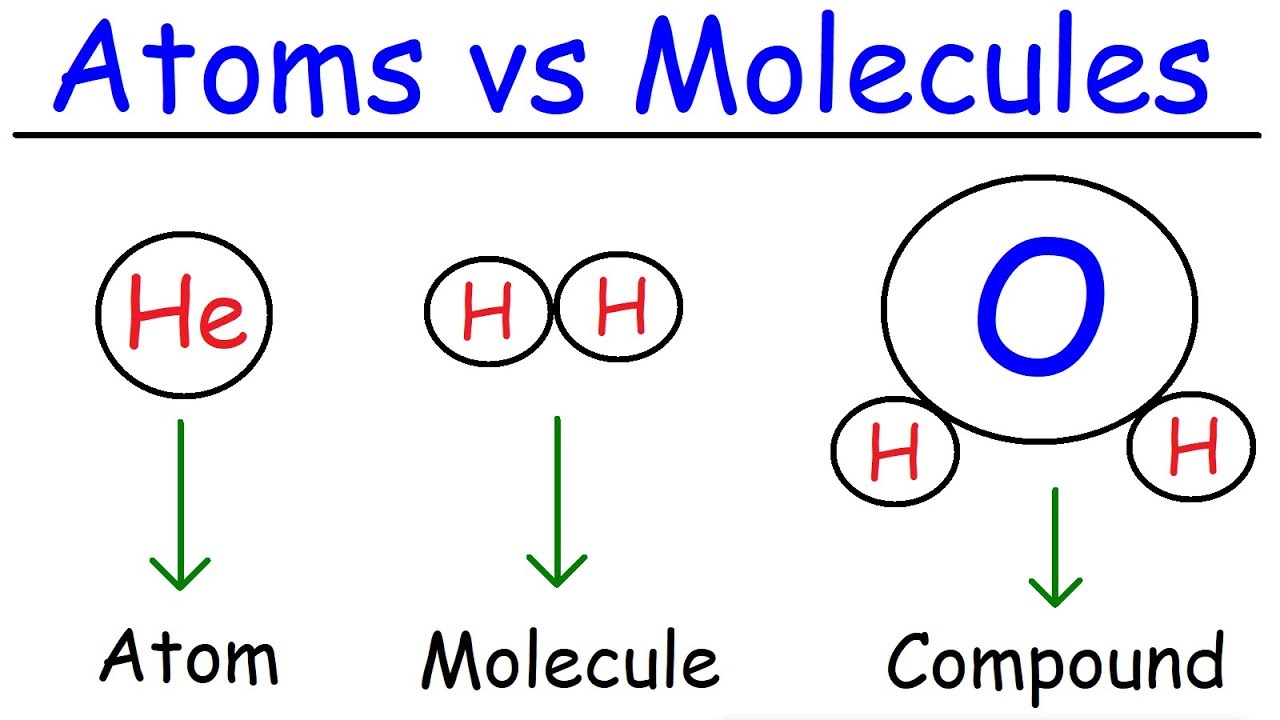
- 🔬 Molecules are defined as two or more atoms held together by chemical bonds.
- 🌐 The term 'molecule' can have different meanings across various scientific fields, but commonly refers to multiple atoms.
- 🧪 Homonuclear molecules are composed of a single chemical element, such as oxygen molecules.
- 🌊 Heterogeneous molecules, like water, consist of different elements, in this case, two hydrogen atoms and one oxygen atom.
- ⚛️ Molecules are neutral and do not carry a charge, distinguishing them from ions.
- 🔍 Molecular size can vary greatly, with most molecules being too small to see without magnification, but some polymers can be macroscopic.
- 🧬 Examples of macroscopic molecules include biopolymers like DNA, which can be seen with the naked eye.
- 🤝 Molecules are formed through covalent bonding, where atoms share electrons, or ionic bonding, where electrons are transferred.
- 💡 Covalent bonds involve a stable balance of forces resulting from electron sharing between atoms.
- ⚡ Ionic bonds occur when one atom transfers valence electrons to another to complete its outer electron shell.
- 📚 The study of molecules relies on quantum mechanics and is crucial for understanding chemical bonds.
- 💭 Philosophically, a molecule is not a fundamental entity but a conceptual tool for observing atomic interactions.
Q & A
What is the basic definition of a molecule?
-A molecule is defined as two or more atoms held together by a chemical bond.
How is the term 'molecule' used in the kinetic theory of gases?
-In the kinetic theory of gases, 'molecule' is a loose term used to refer to any gaseous particle, regardless of its composition.
What are homonuclear molecules?
-Homonuclear molecules are those composed of a single chemical element, such as oxygen molecules (O2).
What is the difference between homonuclear and heteronuclear molecules?
-Homonuclear molecules consist of atoms of the same element, while heteronuclear molecules are composed of different elements, like water (H2O).
What charge do molecules typically have?
-Molecules are typically neutral and have no charge, which distinguishes them from ions.
Can molecules be seen with the naked eye?
-Most molecules are too small to be seen with the naked eye, but some large molecules, like those of many polymers, can reach macroscopic sizes and be visible.
What are the types of bonds that can hold atoms together in a molecule?
-Atoms in a molecule can be held together by covalent bonding, ionic bonding, or metallic bonding. The video focuses on covalent and ionic bonding.
What is a covalent bond?
-A covalent bond is formed when two atoms share electrons between them, creating a stable balance of attractive and repulsive forces.
How is an ionic bond different from a covalent bond?
-An ionic bond is formed when valence electrons are transferred from one atom to another to complete the outer electron shell, unlike covalent bonds which involve electron sharing.
What field of study is essential for understanding the chemical bond?
-The study of molecules by molecular physics and theoretical chemistry, largely based on quantum mechanics, is essential for understanding chemical bonds.
What is the philosophical view on the concept of a molecule?
-Philosophically, a molecule is not a fundamental entity but a scientist's way of making a useful observation about atomic scale interactions.
Outlines

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифMindmap

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифKeywords

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифHighlights

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифTranscripts

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифПосмотреть больше похожих видео

What Is a Molecule?

6.1 Introduction to Chemical Bonding

What's the Difference between an Atom and a Molecule?

Difference between an Atom, a Molecule and a Compound

GCSE Chemistry - Differences Between Compounds, Molecules & Mixtures #3
Elements, Atoms, Molecules, Ions, Ionic and Molecular Compounds, Cations vs Anions, Chemistry
5.0 / 5 (0 votes)
