الروابط الكيميائية الجزء 2 ( الدرس الأول الوحدة الرابعة الفصل الثاني) / علوم الصف الثامن
Summary
TLDRThis lesson explains covalent and ionic bonds in chemistry, focusing on how atoms share or transfer electrons to achieve stable outer energy levels. It illustrates covalent bonding with hydrogen and chlorine examples, showing how shared electrons create weak yet essential molecular bonds. The video also compares physical properties of ionic and covalent compounds, highlighting differences in melting and boiling points, electrical conductivity, and bond strength. Ionic compounds exhibit high melting points and conduct electricity due to charged ions, while covalent compounds have lower melting points and generally do not conduct electricity. Clear visual examples and tables support comprehension of these fundamental chemical concepts.
Takeaways
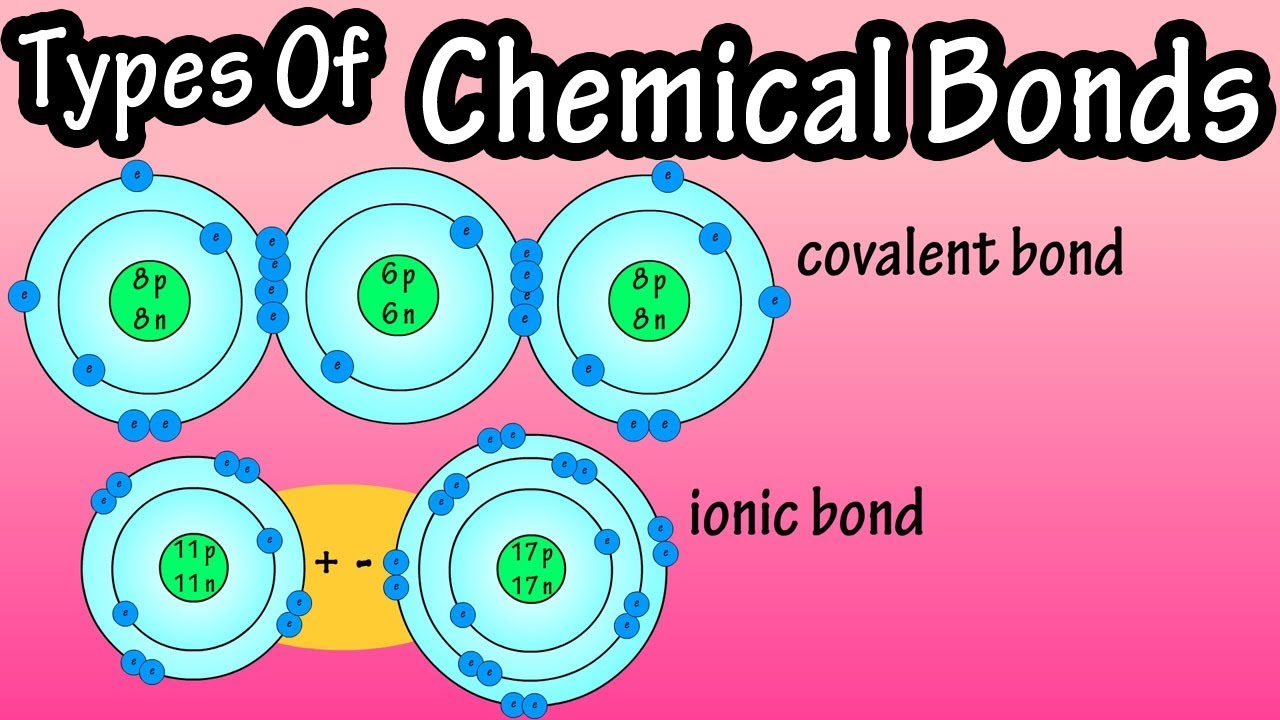
- 😀 The concept of covalent bonding is introduced, where atoms share electrons to achieve stability.
- 😀 In covalent bonds, atoms don't lose or gain electrons but instead share them, leading to a stable electron configuration.
- 😀 The example of H2 (hydrogen molecule) illustrates how two hydrogen atoms share their single electrons to form a stable bond.
- 😀 The covalent bond is described as weak compared to ionic bonds, but still important in chemistry.
- 😀 The role of electron sharing in achieving stable electron shells is emphasized, as shown in H2 and Cl2 molecules.
- 😀 Covalent bonds can form between non-metals, with atoms sharing electrons to complete their outer shells.
- 😀 The concept of electron sharing is explained using examples such as H2O (water molecule), where hydrogen and oxygen atoms share electrons to form covalent bonds.
- 😀 Physical properties of ionic compounds (e.g., high melting and boiling points) are explained due to the strong attraction between ions.
- 😀 In contrast, covalent compounds have lower melting and boiling points due to weaker intermolecular forces.
- 😀 Ionic compounds conduct electricity when dissolved in water because they contain free-moving ions, while covalent compounds usually do not conduct electricity.
- 😀 A table comparing the physical properties of ionic and covalent compounds, such as melting point, boiling point, and electrical conductivity, is discussed to highlight their differences.
Q & A
What is a covalent bond and how does it form?
-A covalent bond forms when two atoms share their valence electrons to achieve a stable outer electron shell, rather than transferring electrons as in ionic bonds.
How does a hydrogen molecule (H₂) demonstrate a covalent bond?
-In H₂, each hydrogen atom has one electron. They share their electrons, so each atom effectively has two electrons in its outer shell, achieving stability.
How do two chlorine atoms form a covalent bond in Cl₂?
-Each chlorine atom has seven valence electrons and needs one more to complete its octet. They share one electron each, creating a covalent bond and completing both outer shells.
Explain the covalent bonding in a water molecule (H₂O).
-Oxygen has six valence electrons and needs two more to complete its octet. It forms covalent bonds by sharing one electron each with two hydrogen atoms, which also complete their outer shells.
Why are covalent bonds considered weaker than ionic bonds?
-Covalent bonds involve shared electrons between atoms, which create weaker attractions compared to the strong electrostatic forces between ions in ionic bonds.
What are the physical properties of ionic compounds?
-Ionic compounds typically have high melting and boiling points due to strong electrostatic attractions between ions and can conduct electricity when dissolved in water.
What are the physical properties of covalent compounds?
-Covalent compounds usually have lower melting and boiling points and are mostly poor conductors of electricity because they do not contain free ions.
Why does pure water (H₂O) not conduct electricity?
-Pure water has no free ions; the electrons are shared in covalent bonds and cannot move freely, so it does not conduct electricity.
How does electron sharing differ from electron transfer in chemical bonding?
-In electron sharing (covalent bonding), atoms share electrons to complete their outer shells. In electron transfer (ionic bonding), one atom gives up electrons and another gains them, creating positive and negative ions.
How can the strength of a chemical bond affect the melting and boiling points of a compound?
-Stronger bonds, like ionic bonds, require more energy to break, resulting in higher melting and boiling points. Weaker covalent bonds require less energy, leading to lower melting and boiling points.
Why is color-coding often used when illustrating covalent bonds?
-Color-coding helps visually distinguish which electrons are shared between atoms, making it easier to understand electron distribution and bond formation.
How can you predict whether a compound will be ionic or covalent?
-Compounds formed between metals and non-metals are usually ionic due to electron transfer, while compounds formed between non-metals are usually covalent due to electron sharing.
Outlines

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードMindmap

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードKeywords

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードHighlights

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードTranscripts

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレード関連動画をさらに表示

IKATAN ANTAR ATOM - Bagaimana Atom Dapat Saling Berikatan?
Types Of Chemical Bonds - What Are Chemical Bonds - Covalent Bonds And Ionic Bonds - What Are Ions

Kimia Kelas 10: Jenis-Jenis Ikatan Kimia

¿Por qué se UNEN los ÁTOMOS? / Enlaces químicos ⚡

Chemical Bonding - Ionic vs. Covalent Bonds

The Octet Rule: Help, Definition, and Exceptions
5.0 / 5 (0 votes)
