The role of the kidney in acid-base balance - renal physiology
Summary
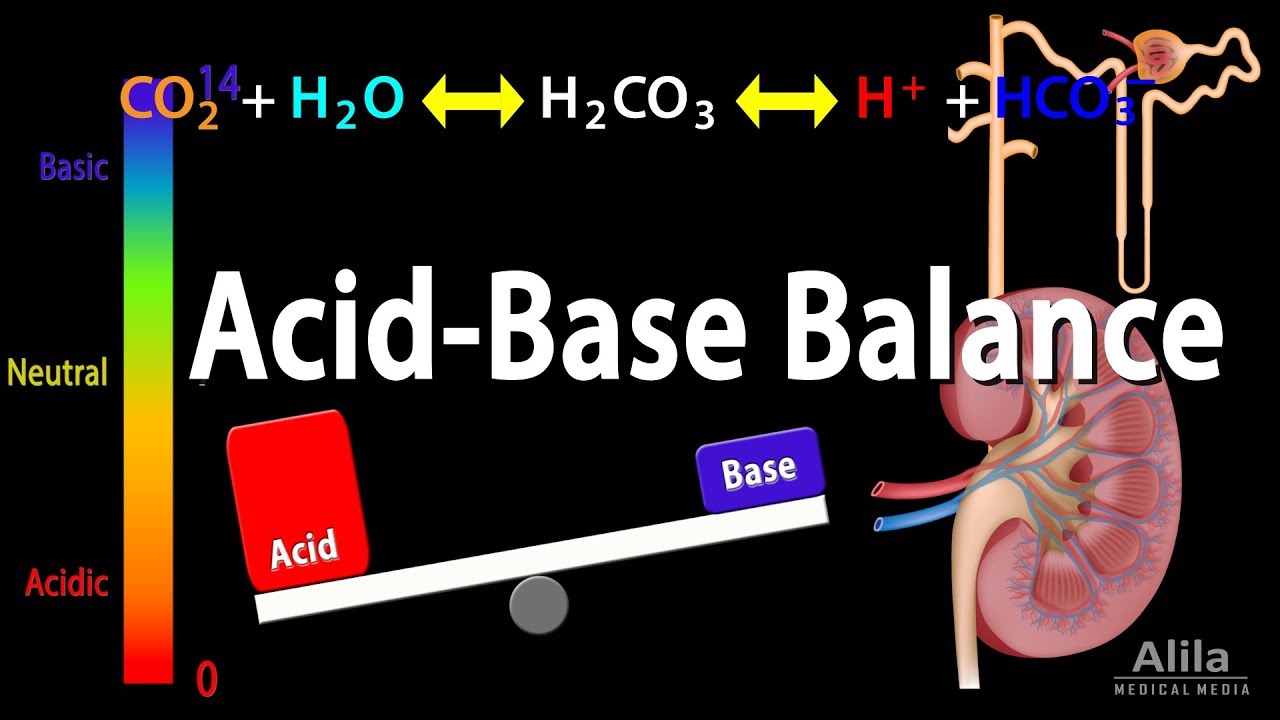
TLDRThe kidneys maintain acid-base balance through two main mechanisms: reabsorbing bicarbonate and secreting hydrogen ions. Blood is filtered in nephrons, where plasma moves into renal tubules. These tubules reabsorb water and electrolytes while leaving waste and acids. Key processes include bicarbonate reabsorption and hydrogen ion secretion, utilizing enzymes like Carbonic anhydrase. Hydrogen ions are expelled through sodium-hydrogen countertransport and ATP-dependent pumps. Buffers like ammonia and phosphate systems prevent urine pH from dropping too low. This complex system ensures the body's pH balance is maintained efficiently.
Takeaways
- 🔄 The kidneys maintain acid-base balance by reabsorbing bicarbonate from the urine back to the blood and secreting hydrogen ions into the urine.
- 🔬 Kidneys filter blood through millions of nephrons, which start with a glomerulus for blood filtration.
- 💧 The renal tubule reabsorbs water and electrolytes, leaving waste products and acids behind.
- 🛠 The renal tubule consists of several segments: the proximal convoluted tubule, the Loop of Henle, the distal convoluted tubule, and the collecting duct.
- 🔬 Brush border cells in the tubules have microvilli to increase surface area for solute reabsorption.
- 🧪 Bicarbonate reabsorption involves binding with hydrogen to form carbonic acid, which is split into water and carbon dioxide by Carbonic anhydrase Type 4.
- 🧪 Carbonic anhydrase Type 2 reverses the reaction, forming bicarbonate and hydrogen, which are then transported back into the blood.
- 💡 Hydrogen ions are secreted into the urine using sodium-hydrogen counter-transport in the proximal tubule and ATP-powered pumps in the distal tubule and collecting ducts.
- 🛡 The ammonia buffer system uses ammonia from amino acid breakdown to combine with hydrogen, forming ammonium which helps prevent urine pH from dropping too low.
- 🔶 The phosphate buffer system uses monohydrogen phosphate to bind hydrogen ions, forming dihydrogen phosphate which is excreted in urine.
- 🩺 Kidneys play a crucial role in maintaining the blood's pH balance through bicarbonate reabsorption and hydrogen ion secretion using various buffer systems.
Q & A
How do kidneys maintain acid-base balance in the body?
-Kidneys maintain acid-base balance by reabsorbing bicarbonate (HCO3-) from the urine back into the blood and secreting hydrogen ions into the urine.
What are nephrons and what is their function in the kidneys?
-Nephrons are tiny functional units in the kidneys that filter blood. They consist of a glomerulus where blood filtration starts and renal tubules that reabsorb necessary substances and leave waste products behind.
Describe the process of blood filtration in the kidneys.
-Blood enters the kidneys and is distributed to nephrons. In the glomerulus, about one-fifth of the plasma is filtered into the renal tubule, which then reabsorbs water and electrolytes while leaving waste products and acids.
What role do brush border cells play in the renal tubules?
-Brush border cells line the renal tubules and have microvilli on their apical surface to increase surface area for solute reabsorption. They help reabsorb bicarbonate and other solutes back into the blood.
Explain the bicarbonate reabsorption process in the proximal convoluted tubule.
-In the proximal convoluted tubule, bicarbonate binds to hydrogen ions to form carbonic acid, which is then split into water and carbon dioxide by Carbonic anhydrase Type 4. These diffuse into cells and are converted back to bicarbonate and hydrogen ions by Carbonic anhydrase Type 2.
How are hydrogen ions secreted into the urine?
-Hydrogen ions are secreted into the urine through two mechanisms: sodium-hydrogen counter transport in the proximal tubule and a hydrogen ATPase pump in the distal tubule and collecting ducts.
Why can't the urine pH drop below 4.5?
-The urine pH can't drop below 4.5 because the tubules cannot maintain such a low pH. To prevent this, the urine uses chemical buffers like the ammonia and phosphate buffer systems to bind hydrogen ions.
What is the ammonia buffer system and how does it work?
-The ammonia buffer system involves the breakdown of amino acids like glutamine into ammonia (NH3) in the proximal convoluted tubule cells. Ammonia combines with hydrogen ions to form ammonium (NH4+), which is excreted in the urine, helping to remove hydrogen ions from the body.
How does the phosphate buffer system help in acid-base balance?
-The phosphate buffer system uses monohydrogen phosphate (HPO4 2-) to bind with hydrogen ions, forming dihydrogen phosphate (H2PO4 -). This process helps to neutralize hydrogen ions and maintain urine pH.
Summarize how the kidneys contribute to pH balance in the blood.
-The kidneys maintain pH balance by reabsorbing bicarbonate ions in the proximal convoluted tubule and secreting hydrogen ions in the proximal and distal convoluted tubules and collecting ducts. The ammonia and phosphate buffer systems help to manage excess hydrogen ions in the urine.
Outlines

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードMindmap

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードKeywords

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードHighlights

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードTranscripts

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレード関連動画をさらに表示
Acid Base Balance, Animation.

Acid Base Physiology | Part One | Basics | Buffers | Renal Physiology

Acid Base Physiology | Part Three | Renal Regulation | Acidification of Urine | Renal Physiology

Acid-base balance: The physiology

Bicarbonate - The Primary Buffer

Metabolic alkalosis - causes, symptoms, diagnosis, treatment, pathology
5.0 / 5 (0 votes)
