Photoelectric Effect, Work Function, Threshold Frequency, Wavelength, Speed & Kinetic Energy, Electr
Summary
TLDRThis educational video delves into the photoelectric effect, explaining how light of a certain frequency can eject electrons from a metal surface. It covers the concept of threshold frequency, the work function, and how to calculate it using Planck's constant. The script provides step-by-step solutions for determining the kinetic energy and speed of ejected electrons, using given wavelengths and work functions of different metals. It also demonstrates conversions between electron volts and joules, and calculates maximum wavelengths for electron ejection from potassium and calcium metals.
Takeaways
- 🌞 The photoelectric effect occurs when light of a certain frequency shines on a metal, causing electrons to be ejected if the light's energy matches or exceeds the metal's work function.
- 🔍 Light with a lower frequency, such as red light, generally doesn't have enough energy to eject electrons from a metal surface, regardless of its intensity.
- 🔵 Higher frequency light, like blue light, can eject electrons from a metal surface, and increasing its intensity can lead to more electrons being ejected.
- ⚡ The work function (W) of a metal is the minimum energy required to remove an electron from the metal's surface and can be calculated using Planck's constant (h) and the threshold frequency (ν₀).
- 📐 The threshold frequency can be found using the formula W = h * ν₀, where W is given and h is Planck's constant.
- 🚀 The kinetic energy of an ejected electron is the difference between the energy of the incident photon and the work function of the metal.
- 📐 The energy of a photon can be calculated using the formula E_photon = h * c / λ, where c is the speed of light and λ is the wavelength of the light.
- 🌈 The speed of an ejected electron can be determined using the kinetic energy and the mass of the electron with the formula KE = 1/2 * m * v².
- 🌌 The maximum wavelength of light that can free an electron from a metal can be found by rearranging the work function equation to solve for wavelength.
- 🔋 The work function of a metal can also be expressed in electron volts (eV), where 1 eV equals 1.6 × 10⁻¹⁹ joules.
- 🛠 To find the speed of an electron, use the kinetic energy formula and solve for velocity, considering the mass of the electron in kilograms.
Q & A
What is the photoelectric effect?
-The photoelectric effect is a phenomenon where electrons are ejected from a metal surface when it is exposed to light of a certain frequency. If the light's frequency is high enough, the energy of the photons can be transferred to the electrons, giving them enough kinetic energy to escape from the metal's atoms.
Why does the frequency of light matter in the photoelectric effect?
-The frequency of light determines whether the photons have enough energy to eject electrons from the metal. If the frequency is too low, even if the light's intensity is high, the electrons will not be ejected because each photon does not carry enough energy.
What is the relationship between light intensity and the number of ejected electrons?
-Once the threshold frequency is surpassed, increasing the light's intensity can increase the number of photons, which in turn can eject more electrons from the metal surface. However, if the threshold frequency is not met, increasing intensity will not affect electron ejection.
What is the work function in the context of the photoelectric effect?
-The work function, often denoted as Φ (Phi), is the minimum energy required to remove an electron from a metal surface. It is typically given in joules and can be calculated using Planck's constant and the threshold frequency.
How can you calculate the threshold frequency of a metal given its work function?
-The threshold frequency can be calculated using the formula for the work function: Φ = h × (threshold frequency), where h is Planck's constant. By rearranging the formula, you can solve for the threshold frequency: threshold frequency = Φ / h.
What is the kinetic energy of an ejected electron?
-The kinetic energy of an ejected electron is the difference between the energy of the incident photon and the work function required to eject the electron. If the photon's energy is greater than the work function, the excess energy becomes the electron's kinetic energy.
How do you find the speed of an electron using its kinetic energy?
-The speed of an electron can be found using the kinetic energy formula: KE = 0.5 × m × v^2, where KE is the kinetic energy, m is the mass of the electron, and v is the speed. By rearranging the formula and solving for v, you can find the electron's speed.
What is the maximum wavelength of light that can free an electron from potassium metal?
-The maximum wavelength can be found by rearranging the work function formula to solve for wavelength: wavelength = (h × c) / Φ, where h is Planck's constant, c is the speed of light, and Φ is the work function in joules. For potassium, with a work function of 2.3 eV, the maximum wavelength is approximately 540 nm.
How can you convert the work function from kilojoules per mole to electron volts?
-To convert the work function from kilojoules per mole to electron volts, first convert kilojoules to joules by multiplying by 1000, then divide by Avogadro's number to get the energy per photon in joules, and finally divide by 1.6 × 10^-19 joules to convert to electron volts.
What is the significance of Avogadro's number in converting work function to electron volts?
-Avogadro's number is used to convert the work function from a bulk scale (per mole) to an individual scale (per photon). Since one mole of any substance contains Avogadro's number of entities, dividing the work function per mole by this number gives the work function per individual photon.
How does the color of light relate to its ability to eject electrons in the photoelectric effect?
-The color of light is related to its frequency, with blue light having a higher frequency than red light. Higher frequency light, such as blue or violet, has enough energy to eject electrons from certain metals, while lower frequency light, like red, usually does not.
Outlines

Dieser Bereich ist nur für Premium-Benutzer verfügbar. Bitte führen Sie ein Upgrade durch, um auf diesen Abschnitt zuzugreifen.
Upgrade durchführenMindmap

Dieser Bereich ist nur für Premium-Benutzer verfügbar. Bitte führen Sie ein Upgrade durch, um auf diesen Abschnitt zuzugreifen.
Upgrade durchführenKeywords

Dieser Bereich ist nur für Premium-Benutzer verfügbar. Bitte führen Sie ein Upgrade durch, um auf diesen Abschnitt zuzugreifen.
Upgrade durchführenHighlights

Dieser Bereich ist nur für Premium-Benutzer verfügbar. Bitte führen Sie ein Upgrade durch, um auf diesen Abschnitt zuzugreifen.
Upgrade durchführenTranscripts

Dieser Bereich ist nur für Premium-Benutzer verfügbar. Bitte führen Sie ein Upgrade durch, um auf diesen Abschnitt zuzugreifen.
Upgrade durchführenWeitere ähnliche Videos ansehen

Física moderna: Efeito Fotoelétrico | Física

3.3.2 - Radiação eletromagnética: Teoria Quântica - Efeito Fotoelétrico (Einstein)

Efeito Fotoelétrico - O Nobel de Einstein

Dual Nature of Radiation & Matter in 10 mins 😱🔥 Ch 11 Physics Class 12 Boards 2022-23 Score 95+
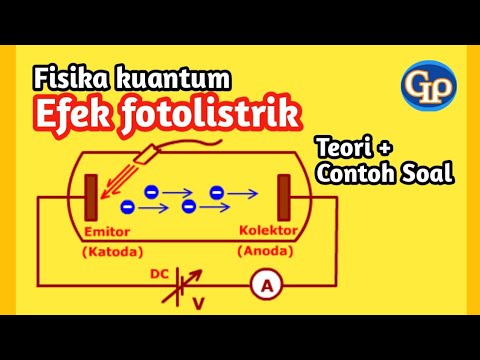
F287- Efek fotolistrik fisika kuantum : Teori plus contoh soal

Photoelectric Effect Theory Lesson
5.0 / 5 (0 votes)
