Stoikiometri Senyawa • Part 1: Kadar Zat - Persen Massa, Persen Volume, bpj / ppm
Summary
TLDRThis educational video from Jendela Sains explores stoichiometry and the concept of substance concentration in high school chemistry. It explains three key ways to express concentration: mass percent, volume percent, and parts per million (PPM), detailing their formulas and applications. The video provides step-by-step examples, calculating the mass of solutes in solutions, maximum allowable concentrations in drinking water, and molarity of concentrated sulfuric acid. Practical tips and a quick formula for molarity are included to simplify calculations, making complex stoichiometry concepts accessible and engaging for students seeking to master the fundamentals of chemical concentrations.
Takeaways
- 😀 Kadar zat refers to the concentration of a substance in a sample, which can be expressed using different methods such as mass percent, volume percent, and parts per million (PPM).
- 😀 Mass percent (%m) measures the mass of a substance in 100 grams of a sample, and the formula is: (Mass of Substance / Mass of Sample) * 100%.
- 😀 Volume percent (%v) calculates the volume of a substance in 100 ml of a solution, often used for liquid solutions or solutes.
- 😀 Parts per million (PPM) expresses the concentration of a substance as mass or volume per one million parts of the sample.
- 😀 The formula for PPM is: (Mass of Substance / Mass of Sample) * 10^6 or (Volume of Substance / Volume of Sample) * 10^6.
- 😀 Mass percent and volume percent are commonly used in chemistry to determine the concentration of substances in mixtures and solutions.
- 😀 PPM is useful when measuring extremely small concentrations, such as pollutants in water or air.
- 😀 In a sample of limestone, if 100 grams contain 70 grams of calcium carbonate, the mass percent of calcium carbonate is 70%.
- 😀 A sample problem showed how to calculate the mass of a solute (CH₃COOH) in a solution with known concentration, volume, and density using mass percent.
- 😀 The video also demonstrated how to calculate the maximum allowable concentration of a substance, such as iron in drinking water, using PPM and density.
- 😀 Molarity and molality are related concepts that also measure concentration, and can be calculated by converting units like volume into liters or using known formulas.
Q & A
What is meant by 'kadar zat' in chemistry?
-'Kadar zat' refers to the concentration or amount of a substance in a mixture or solution. It can be expressed in terms of mass percent, volume percent, parts per million (ppm), or molarity.
How is mass percent (% m/m) calculated?
-Mass percent is calculated by dividing the mass of the solute by the total mass of the sample and multiplying by 100%: % m/m = (mass of solute / mass of sample) × 100%.
What is the difference between percent mass and percent volume?
-Percent mass (% m/m) measures the mass of the solute relative to the total mass of the sample, while percent volume (% v/v) measures the volume of the solute relative to the total volume of the solution, commonly used for liquid mixtures.
How do you calculate parts per million (ppm) for a substance?
-PPM is calculated as the mass of the substance divided by the mass of the sample, multiplied by one million: ppm = (mass of substance / mass of sample) × 10^6. It can also be expressed based on volume if appropriate.
How can you determine the mass of a solute in a solution given percent mass and solution volume?
-First, calculate the mass of the solution using its volume and density (mass = density × volume). Then apply the mass percent formula: mass of solute = % m/m × mass of solution / 100%.
What is the relationship between molarity, mass of solute, and volume of solution?
-Molarity (M) is the number of moles of solute divided by the volume of the solution in liters: M = n / V. If the solute's mass is known, the number of moles n can be calculated as n = mass / molar mass.
How do you calculate molarity if you only know the solution's density and percent mass?
-Use the formula M = (density × 10 × % mass) / molar mass. This converts density and percent mass into moles per liter of solution efficiently.
In the given example, how much CH3COOH is dissolved in 200 ml of 25% m/m solution with a density of 1.05 g/ml?
-The mass of the solution is 200 ml × 1.05 g/ml = 210 g. The mass of CH3COOH is then (25/100) × 210 g = 52.5 g.
What is the maximum mass of iron allowed in a 200 ml glass of drinking water with 0.28 ppm Fe?
-Mass of water = 200 g. Mass of Fe = (0.28 / 10^6) × 200 g = 5.6 × 10^-5 g. This is the maximum allowed iron content.
How do you calculate molarity for concentrated H2SO4 given 98% mass and density 1.8 g/ml?
-Assume 100 g solution: mass of H2SO4 = 98 g. Volume of solution = 100 g / 1.8 g/ml ≈ 55.6 ml = 0.0556 L. Moles of H2SO4 = 98 g / 98 g/mol = 1 mol. Molarity = 1 mol / 0.0556 L ≈ 18 M.
Outlines

Dieser Bereich ist nur für Premium-Benutzer verfügbar. Bitte führen Sie ein Upgrade durch, um auf diesen Abschnitt zuzugreifen.
Upgrade durchführenMindmap

Dieser Bereich ist nur für Premium-Benutzer verfügbar. Bitte führen Sie ein Upgrade durch, um auf diesen Abschnitt zuzugreifen.
Upgrade durchführenKeywords

Dieser Bereich ist nur für Premium-Benutzer verfügbar. Bitte führen Sie ein Upgrade durch, um auf diesen Abschnitt zuzugreifen.
Upgrade durchführenHighlights

Dieser Bereich ist nur für Premium-Benutzer verfügbar. Bitte führen Sie ein Upgrade durch, um auf diesen Abschnitt zuzugreifen.
Upgrade durchführenTranscripts

Dieser Bereich ist nur für Premium-Benutzer verfügbar. Bitte führen Sie ein Upgrade durch, um auf diesen Abschnitt zuzugreifen.
Upgrade durchführenWeitere ähnliche Videos ansehen

Fluida Statis • Part 7: Viskositas, Hukum Stokes, Kecepatan Terminal

Hakikat Ilmu Kimia Kelas 10 • Part 1: Hakikat dan Peran Ilmu Kimia dalam Kehidupan
KONSEP MOL - KIMIA - MATERI UTBK SBMPTN DAN SIMAK UI - UTBK 2022 | SIMAK UI 2022

Concentration of solutions Chemistry
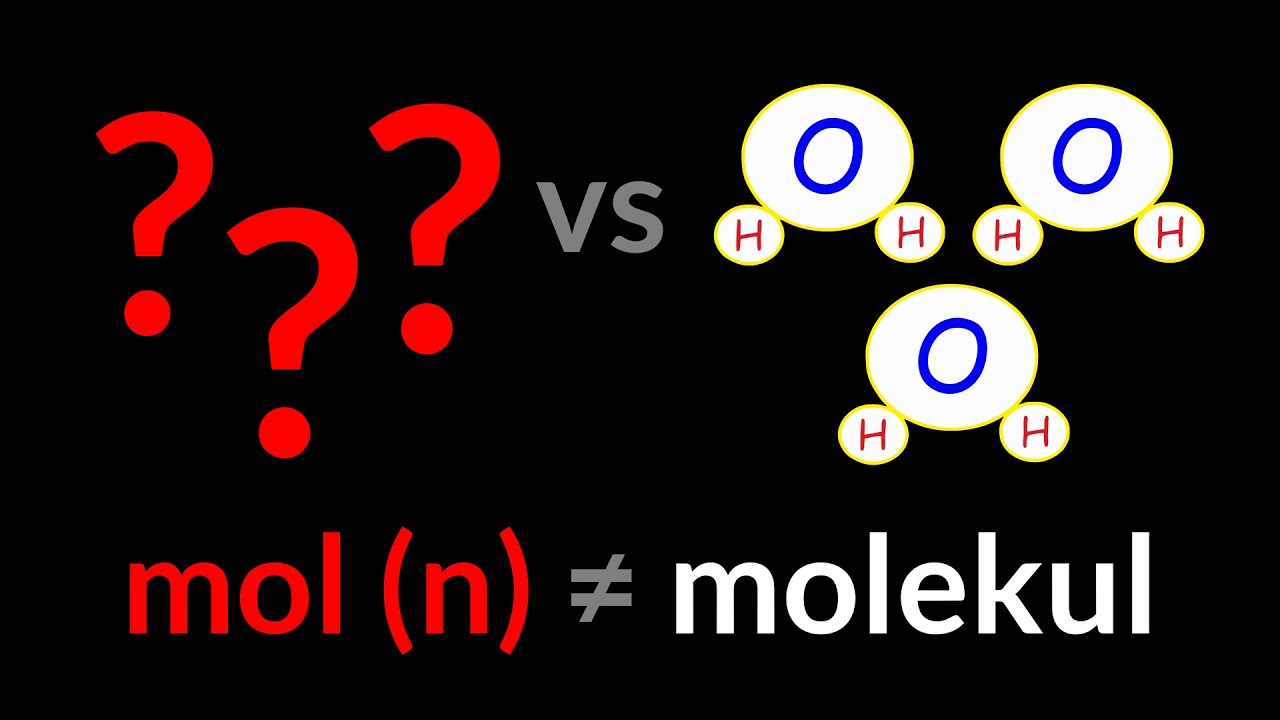
Stoikiometri 1 (mol adalah jumlah)

Matriks Matematika Kelas 11 • Part 23: Menyelesaikan SPLTV dengan Metode Determinan Matriks
5.0 / 5 (0 votes)
