IPA Kelas 9 : Hukum Coulomb dan Cara Menyelesaikan Soal-Soalnya
Summary
TLDRIn this video, the concept of Coulomb's law is explained, focusing on the electric force between charged objects. The law is introduced mathematically, highlighting the direct proportionality to the magnitude of charges and the inverse square relationship to the distance between them. Key units like Coulombs and conversions between micro, milli, and nano units are discussed. Practical examples are provided for calculating electric force, demonstrating how to handle different charge values and distances. The video also covers the effect of varying charges and distances on the resulting force, offering clear steps for applying Coulomb's law in real-world scenarios.
Takeaways
- 😀 Coulomb's Law describes the force between two charged objects, which is directly proportional to the product of their charges and inversely proportional to the square of the distance between them.
- 😀 The formula for Coulomb's Law is: F = k × (Q1 × Q2) / r², where F is the force, Q1 and Q2 are the charges, r is the distance, and k is Coulomb’s constant (9 × 10⁹ N·m²/C²).
- 😀 Electric force is stronger when the charges are larger and weaker when the distance between the charges increases.
- 😀 The unit of electric charge is Coulomb (C), but smaller units like microCoulombs (µC) and nanoCoulombs (nC) are also used. 1 µC = 10⁻⁶ C and 1 nC = 10⁻⁹ C.
- 😀 When working with distances, always ensure units are in meters (m). Convert units such as centimeters (cm) and millimeters (mm) to meters before applying the formula.
- 😀 Power notation is important for Coulomb’s Law calculations. Remember the rules of powers: multiplication adds exponents, division subtracts exponents, and raising a power to another power multiplies the exponents.
- 😀 To calculate forces between charges, ensure you are converting all units correctly before applying the formula. For example, convert microCoulombs to Coulombs and centimeters to meters.
- 😀 A practical example was provided where two charges of 30 µC and -15 µC, separated by 15 cm, were used to calculate the electric force, resulting in a force of 1.8 N.
- 😀 Ratios can be used when charges or distances change. For example, if charges double and distances double, the force will increase by a factor related to these changes.
- 😀 Coulomb's Law can be used to compare the effects of changes in charges or distances. For example, if the charges change to +2Q and -4Q, and the distance doubles, the new force was calculated to be 2 N.
Q & A
What is Coulomb's Law?
-Coulomb's Law describes the electrostatic force between two point charges. It states that the magnitude of the electric force is directly proportional to the product of the magnitudes of the charges and inversely proportional to the square of the distance between them.
What is the formula for Coulomb's Law?
-The formula for Coulomb's Law is: F = k * (Q1 * Q2) / r^2, where F is the electric force, k is Coulomb's constant (9 × 10^9 N·m²/C²), Q1 and Q2 are the charges, and r is the distance between the charges.
What is Coulomb's constant, and what is its value?
-Coulomb's constant, represented by k, is a proportionality factor in Coulomb's Law. Its value is 9 × 10^9 N·m²/C² in a vacuum.
How does the electric force change when the distance between two charges is doubled?
-According to Coulomb's Law, the electric force is inversely proportional to the square of the distance. So, when the distance between two charges is doubled, the force decreases by a factor of 4.
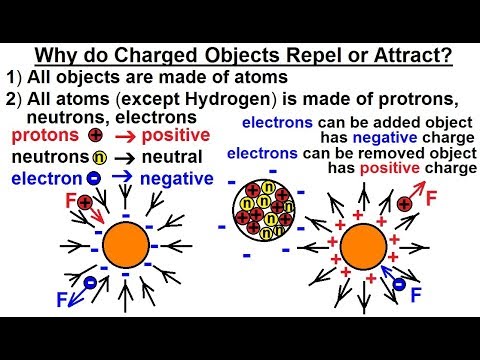
How does Coulomb's Law explain the attraction or repulsion between charged objects?
-Coulomb's Law states that like charges repel each other, and opposite charges attract each other. The force is positive for repulsion and negative for attraction, depending on the nature of the charges.
How do units of Coulomb's Law work, especially with charges and distance?
-In Coulomb's Law, charges are measured in Coulombs (C), distance in meters (m), and the force is measured in Newtons (N). Units of charges like microcoulombs (µC), nanocoulombs (nC), and millicoulombs (mC) need to be converted to Coulombs for calculations.
What is the significance of the dielectric constant (k) in Coulomb's Law?
-The dielectric constant (k) determines how much the electric force is affected by the medium between the charges. In a vacuum, it is 9 × 10^9 N·m²/C², but in other media, it can vary based on the material's properties.
What happens when charges change in a Coulomb's Law problem?
-When charges change in a Coulomb's Law problem, the force can be calculated using ratios. If the charges are increased or decreased, the electric force will change proportionally based on the product of the charges, and the ratio can simplify the calculation.
How do you convert units like microcoulombs to Coulombs for Coulomb's Law calculations?
-To convert units for Coulomb's Law, you multiply by the appropriate power of 10. For example, 1 microcoulomb (µC) = 1 × 10^-6 Coulombs (C), and 1 nanocoulomb (nC) = 1 × 10^-9 Coulombs.
Can you explain the step-by-step calculation of Coulomb's Law with an example?
-Sure! For two charges, Q1 = 30 µC, Q2 = -15 µC, and the distance r = 15 cm, convert the units to Coulombs and meters, then use the formula F = k * (Q1 * Q2) / r^2. After substitution and simplification, you'll get the electric force. In this case, the force is 1.8 N.
Outlines

此内容仅限付费用户访问。 请升级后访问。
立即升级Mindmap

此内容仅限付费用户访问。 请升级后访问。
立即升级Keywords

此内容仅限付费用户访问。 请升级后访问。
立即升级Highlights

此内容仅限付费用户访问。 请升级后访问。
立即升级Transcripts

此内容仅限付费用户访问。 请升级后访问。
立即升级浏览更多相关视频

11TH STD PHYSICS|10.ELECTROSTATICS|LECTURE 03|PRADEEP GIRI SIR

Fisika kelas 12 | Listrik Statis (part 1) Muatan listrik dan gaya coulomb

Tema 01 - A Carga Elétrica e o Spin | Experimentos - Lei de Coulomb
Physics - E&M: Ch 35.1 Coulumb's Law Explained (1 of 28) Why do Charged Objects Repel or Attract?

Listrik Statis, Konsep Hukum Coulomb dan Contoh-contoh Soal

Eduscribe : Fisika (Listrik Statis) Part 1
5.0 / 5 (0 votes)
