Life and the Ocean's Chemical Environment
Summary
TLDRThis video explores the essential role of diffusion and osmosis in the survival of organisms. It details how oxygen is transferred into the blood through diffusion in the lungs of mammals and gills of fish, highlighting their different efficiencies. Additionally, it explains the processes of osmosis in saltwater and freshwater environments and how fish adapt to regulate water and salt in their bodies. The video also touches on active transport and its energy requirements, emphasizing the importance of these biological mechanisms for marine life and ecosystem diversity.
Takeaways
- 🌍 Almost all organisms on Earth require oxygen to survive, which is primarily obtained through diffusion.
- 💨 In humans and other mammals, oxygen diffuses into the blood from the lungs, while carbon dioxide diffuses out.
- 🫁 Human lungs have a 25% efficiency rate in transferring oxygen from the air into the bloodstream.
- 🐟 Fish use gills, which have a surface area ten times greater than their body, allowing them to extract 85% of oxygen from water.
- 🦴 Bony fish and cartilaginous fish differ in their gill structures, with bony fish having an operculum and cartilaginous fish having gill slits.
- 🌊 Diffusion is also crucial for marine autotrophs to absorb nutrients and for other organisms to eliminate waste.
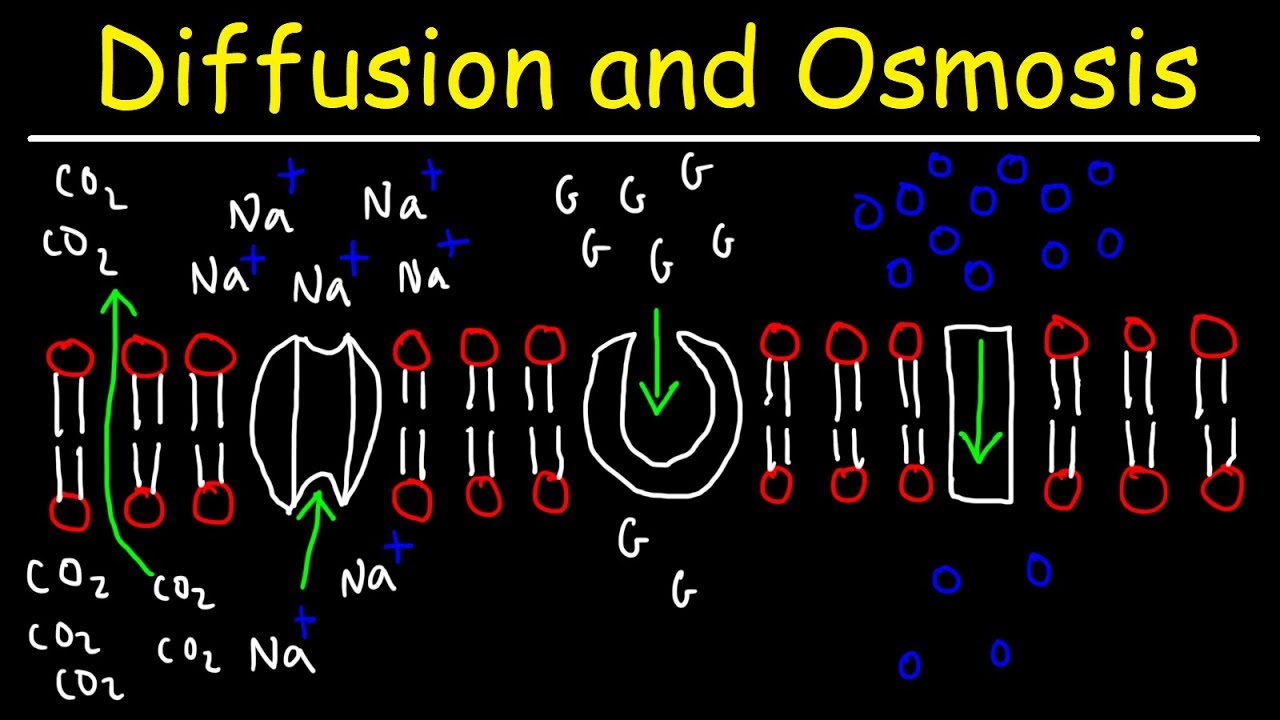
- 🔋 Active transport is an energy-dependent process where molecules move against a concentration gradient, as seen in fat storage.
- 💧 Osmosis occurs when water moves across a boundary toward an area needing dilution, causing cells to either swell or shrink based on the surrounding water's salinity.
- 🐠 Saltwater fish drink seawater and excrete salt, while freshwater fish do not drink water and expel it frequently through urination.
- 🦈 Few fish can handle both saltwater and freshwater environments due to the challenges of osmoregulation, making estuaries less diverse ecosystems.
Q & A
What is diffusion and how does it work in human lungs?
-Diffusion is the process where molecules move freely across boundaries to achieve equal concentrations on both sides. In human lungs, oxygen from the air diffuses into oxygen-poor blood, while carbon dioxide diffuses from the blood into the lungs to be exhaled.
How efficient are human lungs at extracting oxygen from the air?
-Human lungs have an efficiency of 25%, meaning 25% of the available oxygen in the air we breathe is transferred to the bloodstream.
How do fish extract oxygen more efficiently than humans?
-Fish use gills, which have a surface area ten times greater than the fish's body. This allows them to extract up to 85% of the available oxygen from water as it flows across their gill filaments.
What is the difference between bony fish and cartilaginous fish in terms of their gills?
-Bony fish typically have an operculum, a bony covering that protects their gills, while cartilaginous fish, such as sharks, have gill slits instead of an operculum.
What role does diffusion play in marine autotrophs and other simple organisms?
-Diffusion is essential for marine autotrophs like seaweeds and sponges, as it allows them to absorb nutrients and eliminate wastes directly through their outer cell walls.
What is the difference between diffusion and osmosis?
-Diffusion involves molecules moving freely to achieve equilibrium across a boundary, while osmosis specifically involves water molecules moving through a semi-permeable membrane to balance solute concentrations on both sides.
What happens to human skin cells when submerged in fresh water for a long time?
-Water moves into the skin cells in an attempt to dilute the salty fluid inside them, causing the skin to swell and take on a prune-like appearance.
Why does swimming in seawater dehydrate the human body?
-The water inside the body's cells moves out into the saltier seawater to balance the salt concentration, leading to dehydration.
How do saltwater fish regulate osmosis?
-Saltwater fish drink seawater and use specialized cells in their gills to remove excess salt. They excrete this salt through highly salty urine to maintain their internal water balance.
Why are estuaries low in fish diversity?
-Estuaries have low fish diversity because the constant changes in salinity require fish to continuously adapt their osmotic regulation processes, a challenge for many species.
Outlines

此内容仅限付费用户访问。 请升级后访问。
立即升级Mindmap

此内容仅限付费用户访问。 请升级后访问。
立即升级Keywords

此内容仅限付费用户访问。 请升级后访问。
立即升级Highlights

此内容仅限付费用户访问。 请升级后访问。
立即升级Transcripts

此内容仅限付费用户访问。 请升级后访问。
立即升级浏览更多相关视频

Osmoregulation

Mekanisme Transpor Pada Membran Sel || BIOLOGI SMA

How Large Can a Bacteria get? Life & Size 3

2025 ATI TEAS 7 Science Chemistry with Nurse Cheung | Properties of Solutions, Osmosis, Diffusion
Diffusion and Osmosis - Passive and Active Transport With Facilitated Diffusion
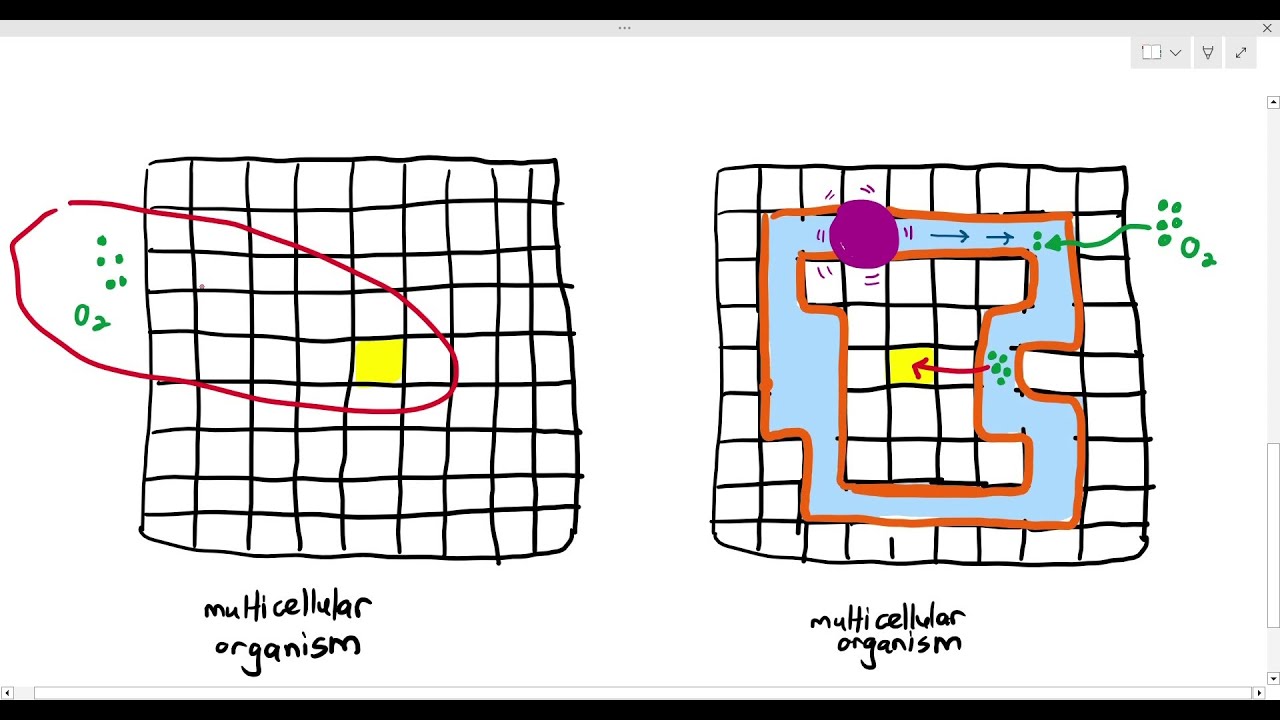
8-1 Why Some Multicellular Organisms Need A Transport System (Cambridge AS & A Level Biology, 9700)
5.0 / 5 (0 votes)
