A Level Chemistry Revision "Electron Configuration"
Summary
TLDRThis educational video script from the 'Three Science' series teaches viewers how to determine electron configurations of elements. It explains the concept of atomic orbitals, subshells, and the energy levels associated with them. The script outlines three key rules for electron assignment: filling the lowest energy orbitals first, allowing two electrons per orbital with opposite spins, and distributing electrons across degenerate orbitals before pairing. Examples are provided, starting with hydrogen and helium, then challenging viewers to determine lithium's configuration and finally nitrogen's, which has seven electrons. The script promises to explore more complex examples and exceptions in a subsequent video.
Takeaways
- 🔬 The video is a chemistry lesson focused on electron configuration in elements.
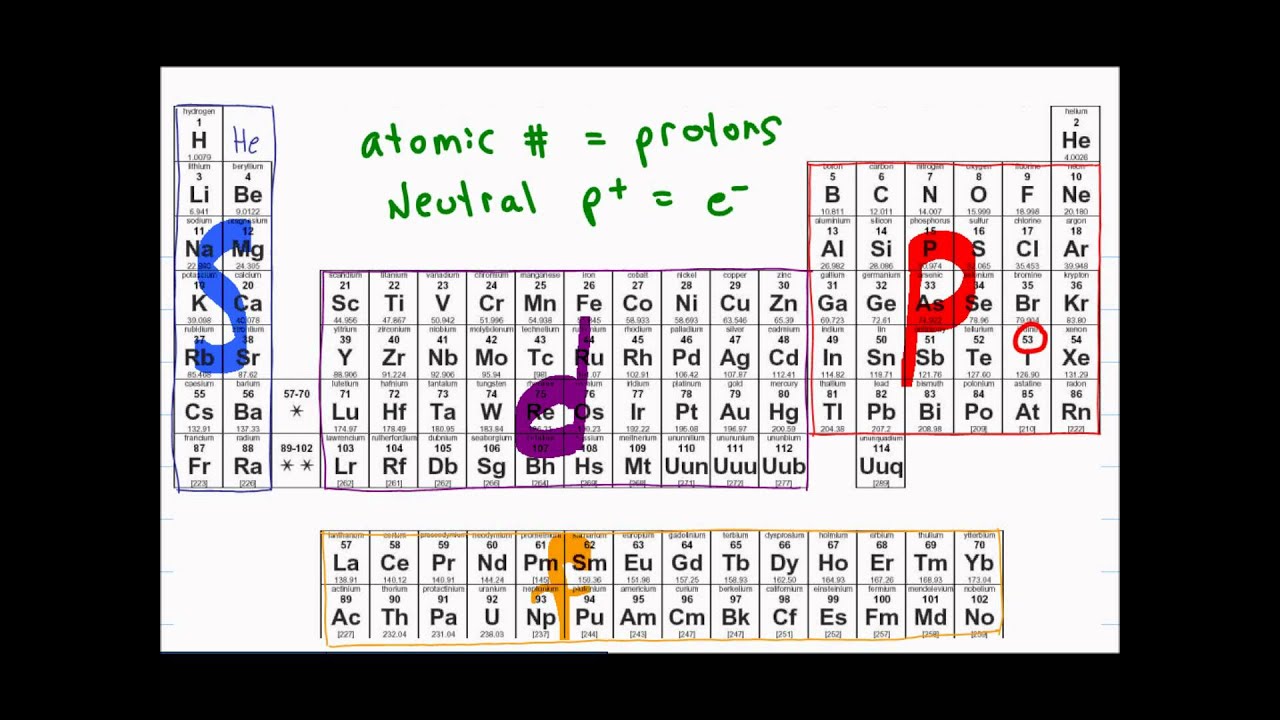
- 🌐 An atomic orbital is a region around the nucleus where up to two electrons with opposite spins can reside.
- 📚 There are different types of orbitals: s, p, d, and f orbitals, each with unique characteristics.
- 📈 Subshells, which are groups of orbitals of the same type in the same shell, have varying energies.
- 📉 The energy of subshells increases as we move away from the nucleus.
- ✅ Three key rules for assigning electrons to orbitals are: fill the lowest energy orbitals first, each orbital can hold two electrons with opposite spins, and distribute electrons into individual orbitals before pairing when energies are the same.
- 💡 The video provides examples of how to assign electrons to orbitals, starting with hydrogen and helium.
- 🚀 Electron configuration is a shorthand notation that represents the distribution of electrons in an atom's subshells.
- 🌿 The example of nitrogen is used to illustrate how to determine electron configuration, which for nitrogen is 1s² 2s² 2p³.
- 🔍 The video concludes with a teaser for the next part, which will cover more complex examples and exceptions to the rules of electron configuration.
Q & A
What is an atomic orbital?
-An atomic orbital is a region around a nucleus where electrons are most likely to be found and can hold up to two electrons with opposite spins.
What are the different types of orbitals mentioned in the script?
-The different types of orbitals mentioned are s, p, d, and f orbitals.
What is a subshell in the context of atomic orbitals?
-A subshell consists of all orbitals of the same type within the same shell, such as the 3d subshell shown in the script.
How does the energy of subshells change as we move away from the nucleus?
-As we move away from the nucleus, the energy of the subshells increases.
What are the three rules for assigning electrons to atomic orbitals?
-The three rules are: 1) Orbitals with the lowest energy are filled first. 2) Up to two electrons with opposite spins can occupy the same orbital. 3) If orbitals have the same energy, electrons are placed in individual orbitals before pairing them.
How is the electron configuration of hydrogen represented in the script?
-The electron configuration of hydrogen is represented as 1s1, indicating one electron in the 1s subshell.
Why do electrons in the same orbital have opposite spins?
-Electrons in the same orbital must have opposite spins due to the Pauli Exclusion Principle, which states that no two electrons in an atom can have the same set of quantum numbers.
What is the electron configuration for helium as described in the script?
-The electron configuration for helium is 1s2, indicating two electrons in the 1s subshell with opposite spins.
How does the script describe the electron configuration for lithium?
-The electron configuration for lithium is 1s2 2s1, showing the first two electrons in the 1s subshell and the third electron in the 2s subshell.
What is the process for assigning the remaining electrons to orbitals in nitrogen as per the script?
-For nitrogen, the first two electrons go into the 1s subshell, the next two into the 2s subshell, and the remaining three electrons go into the 2p subshell, with the electron configuration being 1s2 2s2 2p3.
Why are individual orbitals in the 2p subshell not shown in the electron configuration?
-Individual orbitals in the 2p subshell are not shown in the electron configuration because it only represents the subshells, not the individual orbitals within them.
Outlines

此内容仅限付费用户访问。 请升级后访问。
立即升级Mindmap

此内容仅限付费用户访问。 请升级后访问。
立即升级Keywords

此内容仅限付费用户访问。 请升级后访问。
立即升级Highlights

此内容仅限付费用户访问。 请升级后访问。
立即升级Transcripts

此内容仅限付费用户访问。 请升级后访问。
立即升级浏览更多相关视频

How to Write the Electron Configuration for an Element in Each Block

A Level Chemistry Revision "Electron Configuration and the Periodic Table"

KONFIGURASI ELEKTRON MENURUT TEORI ATOM BOHR & ELEKTRON VALENSI (KIMIA SMA KELAS 10)
Electron Configuration of Atoms + Shortcut Tutorial Video

Cara Mudah Menentukan Periode dan Golongan Kimia Kelas 10

The Aufbau principle | Atomic structure and properties | AP Chemistry | Khan Academy
5.0 / 5 (0 votes)
