IB Chemistry Topic 1 Stoichiometric relationships Topic 1.2 The mole concept SL
Summary
TLDRThis video covers foundational concepts in chemistry, focusing on the mole, conversions between different units, and the mole’s role in calculating volumes, masses, and particle counts. It explores practical problems like converting calories to joules, determining the number of moles in a given mass, and working with atomic and molecular masses. The video also delves into concepts such as percent composition, empirical and molecular formulas, and stoichiometry. Using clear examples, it shows how to convert mass to moles, solve for unknowns, and calculate chemical compositions, making complex topics accessible for learners.
Takeaways
- 😀 Understanding the mole: A mole is a large number used to represent particles like atoms, molecules, or ions in chemistry.
- 😀 Conversion of units: The script emphasizes the importance of converting between different units, such as nanometers to centimeters and calories to joules, using ratios and cross-multiplication.
- 😀 Cross-multiplication method: A useful technique for solving ratio problems, especially in unit conversions, where you can set up equations and cross-multiply to solve for unknown values.
- 😀 Mole calculations: The mole allows you to work with large quantities of particles, and the number of particles can be calculated by multiplying the number of moles by Avogadro's number (6.02 × 10^23).
- 😀 Mass and moles: The molar mass (grams per mole) is essential for converting between the mass of a substance and the number of moles.
- 😀 Empirical vs. molecular formulas: The empirical formula represents the simplest whole number ratio of elements in a compound, while the molecular formula represents the actual number of atoms of each element in a molecule.
- 😀 Percent composition: To calculate the percent composition of an element in a compound, divide the mass of the element by the total mass of the compound and multiply by 100.
- 😀 Unit conversions for gases: In stoichiometry, it's crucial to convert between different phases (solid, liquid, gas) to work with chemical reactions and their respective mole ratios.
- 😀 Atomic and molecular mass terminology: The script stresses the importance of understanding atomic mass (usually given for carbon-12 as 12), molecular mass, and the proper units (grams per mole).
- 😀 Example problems: The script provides several worked examples for converting between mass and moles, calculating molecular formulas, and finding empirical formulas, which reinforce the application of theoretical concepts.
Q & A
What is the importance of converting between different units in chemistry?
-Converting between different units in chemistry is crucial because it ensures consistency in measurements, allows for proper calculation of chemical quantities, and helps when working with different scales, such as nanometers to centimeters or calories to joules.
How can the mole concept be useful in understanding chemical reactions?
-The mole concept helps quantify the number of particles in a sample, making it easier to relate the macroscopic amounts of substances to the microscopic level of atoms, ions, or molecules. It is essential for balancing equations and performing stoichiometric calculations.
How do you convert calories to joules?
-To convert calories to joules, you multiply the number of calories by the conversion factor of 4.18 (since 1 calorie = 4.18 joules). For example, 530 calories = 530 * 4.18 = 2,215 joules.
What is the relationship between the number of atoms and the number of moles in a sample?
-The number of atoms in a sample can be calculated by multiplying the number of moles by Avogadro's number (6.02 x 10^23). For example, 1 mole of H2O contains 3 atoms, and thus 1 mole of H2O contains 1.806 x 10^24 atoms.
What is the difference between the molecular formula and empirical formula?
-The molecular formula shows the exact number of atoms of each element in a molecule, while the empirical formula gives the simplest whole number ratio of the elements in a compound. For example, C6H12O6 is the molecular formula of glucose, while CH2O is its empirical formula.
How do you calculate the molar mass of a compound?
-To calculate the molar mass, you sum the atomic masses of all the elements in the compound, multiplied by the number of atoms of each element. For example, the molar mass of NaCl is the atomic mass of sodium (22.99) plus the atomic mass of chlorine (35.45), which gives 58.44 g/mol.
What is meant by the 'percent composition' of an element in a compound?
-The percent composition of an element in a compound is the percentage by mass of each element in the total mass of the compound. For example, in sulfur dioxide (SO2), the percent composition of sulfur is 50%, calculated by dividing the mass of sulfur by the total molecular mass and multiplying by 100.
What is the formula for converting between mass and moles?
-The formula for converting between mass (m) and moles (n) is n = m / M, where n is the number of moles, m is the mass in grams, and M is the molar mass in g/mol.
How do you determine the empirical formula from percentage composition?
-To determine the empirical formula from percentage composition, assume a 100 g sample, convert the percentages to grams, and then convert those masses to moles. Divide by the smallest number of moles to find the simplest whole number ratio of elements.
How is the molecular formula related to the empirical formula?
-The molecular formula is a whole number multiple of the empirical formula. If the molar mass of the compound is known, you can divide it by the molar mass of the empirical formula to find the multiple and determine the molecular formula.
Outlines

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowMindmap

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowKeywords

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowHighlights

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowTranscripts

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowBrowse More Related Video

Mole Concept Tips and Tricks
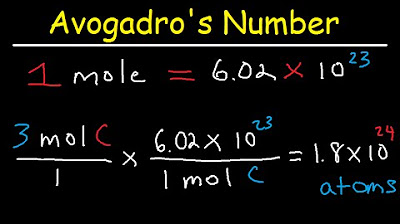
Avogadro's Number, The Mole, Grams, Atoms, Molar Mass Calculations - Introduction
Kelas Kimia : Konsentrasi Larutan (% berat, % volume, ppm / bpj)

11 - Relações entre C, T, d, M Concentração comum, título, densidade e molaridade.

QUANTITATIVE CHEMISTRY - GCSE Chemistry (AQA Topic C3)

konsep mol E2
5.0 / 5 (0 votes)