Ionic vs. Molecular
Summary
TLDRThis educational video script explores the distinction between ionic and molecular (covalent) compounds. It explains that ionic compounds consist of metals and nonmetals, while molecular compounds are made solely of nonmetals. The script uses examples like sodium chloride and sulfur dioxide to illustrate the concepts and highlights key differences, such as how atoms bond through sharing (molecular) or electron theft (ionic). It also touches on the structural differences, with molecular compounds forming distinct molecules and ionic compounds forming lattice structures.
Takeaways
- 🔬 Ionic compounds are formed from metals and nonmetals, while molecular (covalent) compounds are formed from nonmetals only.
- 📚 To determine if a compound is ionic or molecular, refer to the periodic table and check the elements' positions relative to the metal-nonmetal staircase.
- 🌐 Sulfur Dioxide (SO2) is a molecular compound because it's composed of sulfur and oxygen, both nonmetals.
- ⚛️ Sodium Chloride (NaCl) is an ionic compound, consisting of the metal sodium and the nonmetal chlorine.
- 💧 Water (H2O) is a molecular compound despite hydrogen's position on the periodic table; it's an exception and is considered a nonmetal.
- 🧪 Copper (II) Fluoride is an ionic compound, with copper as a metal and fluorine as a nonmetal.
- 🍶 Propanol, composed of carbon, hydrogen, and oxygen, is a molecular compound even though it contains multiple nonmetals.
- 🧂 Lithium Nitrate and Sodium Sulfate are ionic compounds with a metal and multiple nonmetals, forming polyatomic ions.
- 🤝 In molecular compounds, atoms are held together by sharing electrons, forming covalent bonds.
- 🔗 In ionic compounds, atoms come together because one atom steals an electron from another, creating opposite charges that attract each other.
- 🌡️ The structure of compounds differs: molecular compounds form discrete molecules, while ionic compounds form lattice structures.
Q & A
What is the main difference between ionic and molecular compounds?
-The main difference is that ionic compounds are made of metals and nonmetals and are held together by the electrostatic attraction between oppositely charged ions, while molecular or covalent compounds are made of only nonmetals and are held together by shared electrons.
How can you determine if a compound is ionic or molecular by looking at its elements?
-A compound is likely ionic if it is made of metals and nonmetals, and it is likely molecular if it is made of only nonmetals.
What role does the periodic table play in identifying ionic versus molecular compounds?
-The periodic table helps by showing which elements are metals and which are nonmetals. Metals are typically found on one side of the staircase that divides the table, and nonmetals are on the other side.
Why is hydrogen considered a nonmetal despite its position on the periodic table?
-Hydrogen is an exception to the general rule. Even though it is positioned with metals on the periodic table, it is actually a nonmetal.
What is a polyatomic ionic compound and how does it differ from other ionic compounds?
-A polyatomic ionic compound is an ionic compound that contains two or more different nonmetals in its formula. It differs from other ionic compounds in that it has more complex anions or cations that are groups of atoms rather than single atoms.
How are atoms held together in molecular compounds?
-In molecular compounds, atoms are held together by sharing electrons, which is represented by covalent bonds between the atoms.
What is the process by which atoms in ionic compounds stick together?
-In ionic compounds, atoms stick together because one atom steals an electron from another, creating opposite charges that attract each other, similar to how magnets attract.
What is the difference in structure between molecular and ionic compounds?
-Molecular compounds are made of discrete molecules, which are clumps of atoms held together by covalent bonds. Ionic compounds, on the other hand, form lattice structures where the ions are arranged in a regular, repeating pattern.
What happens to ionic and molecular compounds when they dissolve in water?
-Molecular compounds dissolve into individual molecules, while ionic compounds dissociate into their constituent ions.
Why are there no individual clumps of atoms in ionic compounds like there are in molecular compounds?
-In ionic compounds, the atoms are arranged in a continuous lattice structure due to the electrostatic forces between the ions, whereas in molecular compounds, atoms are grouped into discrete molecules by sharing electrons.
What are some physical properties that distinguish ionic from molecular compounds?
-Ionic compounds often have high melting and boiling points due to the strong electrostatic forces, while molecular compounds typically have lower melting and boiling points because the forces between molecules are weaker.
Outlines

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowMindmap

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowKeywords

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowHighlights

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowTranscripts

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowBrowse More Related Video

Tata Nama Senyawa | KIMIA KELAS 10

Introduction to Naming Simple Ionic and Covalent Compounds
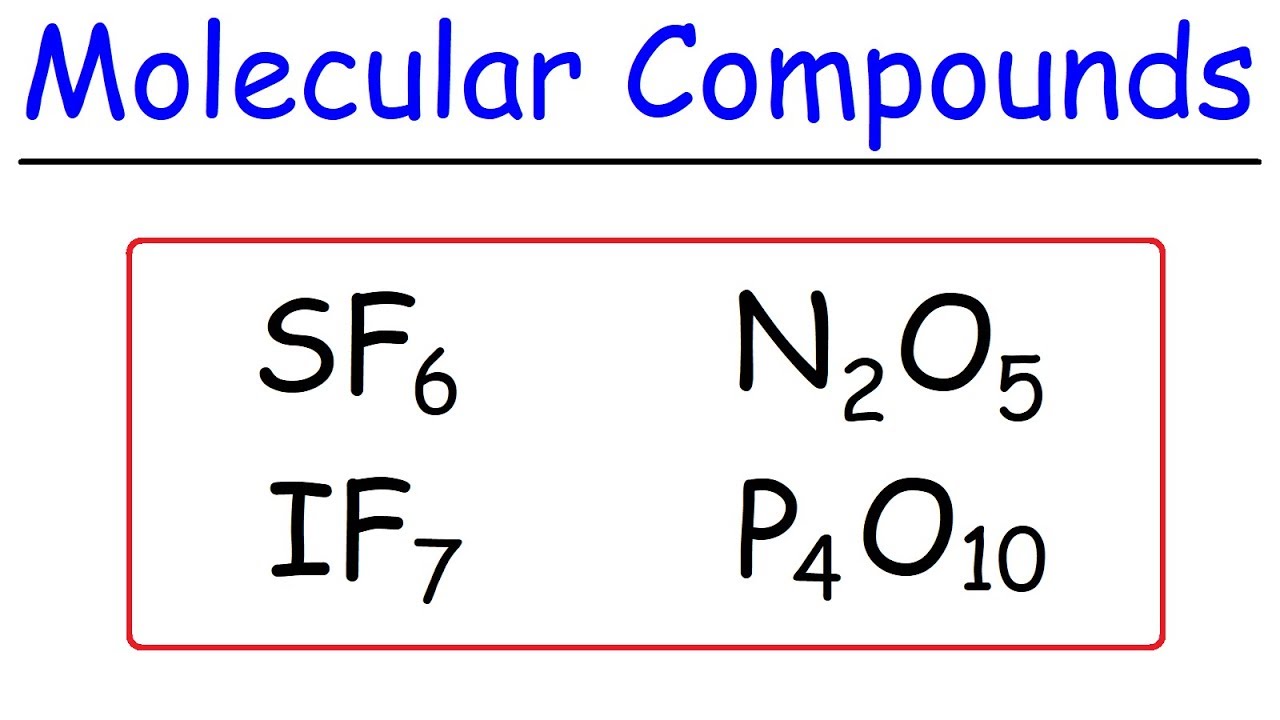
How To Name Covalent Molecular Compounds - The Easy Way!

Soal-soal Ikatan Kimia Kelas 10 dan 11 SMA/MA Pilihan

الروابط الكيميائية الجزء 2 ( الدرس الأول الوحدة الرابعة الفصل الثاني) / علوم الصف الثامن

How Do Atoms Bond - Part 2 | Properties of Matter | Chemistry | FuseSchool
5.0 / 5 (0 votes)