Aufbau Principle (Periodic Table Labels).mp4
Summary
TLDRThis vodcast explains how to label the periodic table to understand electron configurations, focusing on the Aufbau principle. It highlights how electrons fill lower energy orbitals first and introduces labeling sections for s, p, d, and f blocks. The video also covers orbital overlap, particularly the 3d and 4s energy levels, and guides viewers through the proper order for filling orbitals across the periodic table. Finally, it reassures viewers that they don't need to stress over the f-block elements due to their limited role in most chemistry studies.
Takeaways
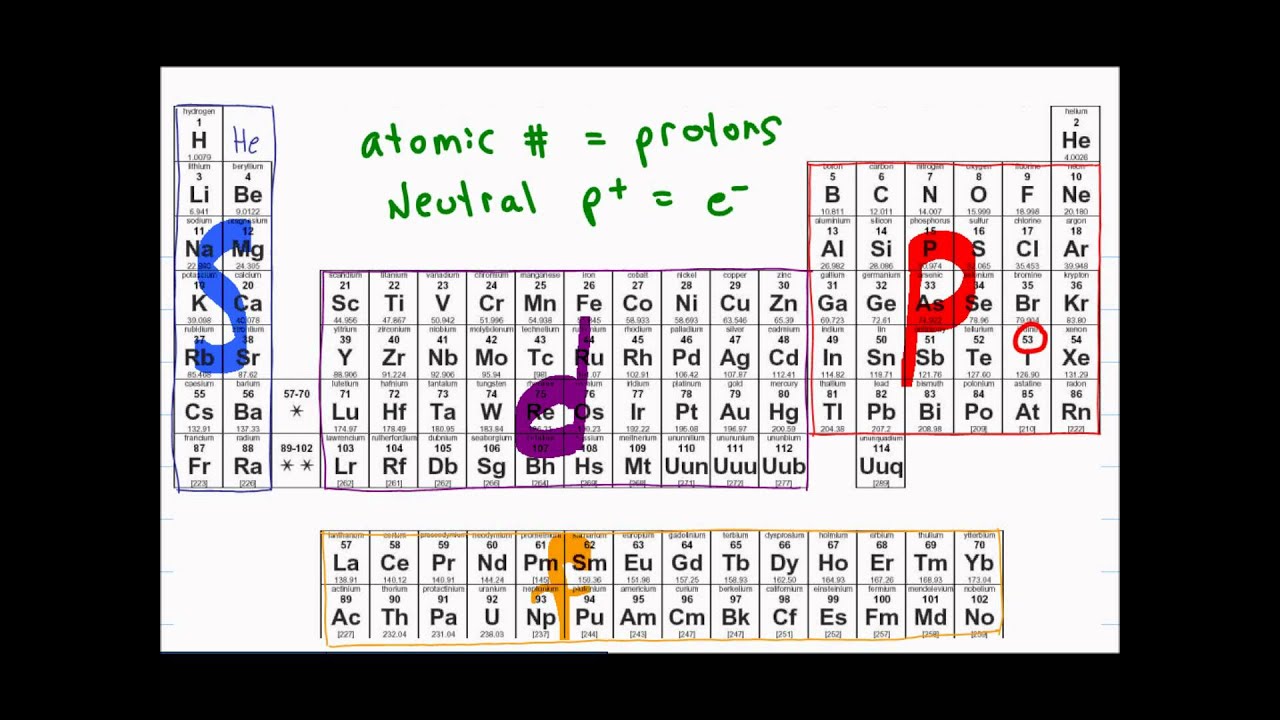
- 📝 The video explains how to label the periodic table with electron configurations using the Aufbau principle.
- 🔋 The Aufbau principle states that electrons fill lower energy levels first and higher energy levels last.
- 🧊 Energy levels overlap starting from the third level, making the order of filling orbitals confusing without labels.
- 🔍 Helium is temporarily considered part of the second group on the periodic table for electron configuration purposes.
- 🔢 The energy levels are labeled 1 through 7, indicating the primary levels where electrons reside.
- 🟧 The periodic table is divided into blocks: the S block (first two columns), P block (right side), D block (middle), and F block (bottom).
- 🪜 The order of electron filling starts at 1s, then 2s, 2p, 3s, 3p, and continues in a specific sequence influenced by energy levels.
- ⚠️ Due to orbital overlap, the 4s orbital fills before the 3d orbital because 4s has lower energy.
- 📉 The F block elements, found in the bottom two rows, have less focus since they are radioactive and not part of typical chemistry studies.
- ✅ The main takeaway is that labeling the periodic table helps identify the correct sequence for electron configurations, which follows the Aufbau principle from left to right and down each row.
Q & A
What is the Aufbau principle?
-The Aufbau principle states that electrons fill orbitals from low energy to high energy, starting with the lowest available energy level before moving to higher ones.
Why is helium penciled in as part of the second group on the periodic table?
-Helium is temporarily penciled into the second group of the periodic table because it helps with organizing electron configurations, even though it belongs to the noble gases in Group 18.
How are energy levels labeled on the periodic table?
-Energy levels on the periodic table are labeled as 1, 2, 3, 4, 5, 6, and 7, corresponding to the principal quantum numbers that describe the different energy levels electrons can occupy.
What does the S block on the periodic table represent?
-The S block on the periodic table represents the first two columns, where all elements have their outermost electrons in S orbitals.
How do you identify the P block on the periodic table?
-The P block includes the elements in Groups 13-18 (excluding helium). It is found on the right side of the periodic table and includes elements whose outermost electrons are in P orbitals.
What is the D block, and where is it located on the periodic table?
-The D block, located in the center of the periodic table, includes transition metals. The outermost electrons of these elements are in D orbitals.
What is the significance of the F block on the periodic table?
-The F block includes the lanthanides and actinides, which are elements located below the main body of the periodic table. These elements have their outermost electrons in F orbitals.
Why does the 4S orbital fill before the 3D orbital?
-Although the 3D orbital belongs to the third energy level, the 4S orbital has a lower energy, so it fills before the 3D orbital due to orbital overlap.
What is the electron filling order for energy levels and sublevels?
-The order is 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p.
Why is less focus placed on the elements in periods 6 and 7, particularly the F block?
-Elements in periods 6 and 7, especially in the F block, are typically radioactive and do not participate in the types of chemistry that are usually studied in detail, so they are less emphasized.
Outlines

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифMindmap

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифKeywords

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифHighlights

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифTranscripts

Этот раздел доступен только подписчикам платных тарифов. Пожалуйста, перейдите на платный тариф для доступа.
Перейти на платный тарифПосмотреть больше похожих видео
Electron Configuration of Atoms + Shortcut Tutorial Video

ELECTRON CONFIGURATION | AUFBAU PRINCIPLE | HUND'S RULE | PAULI EXCLUSION PRINCIPLE | S-P-D-F

The Aufbau principle | Atomic structure and properties | AP Chemistry | Khan Academy

Hund's Rule

KIMIA Kelas 10 - Konfigurasi Elektron | GIA Academy

Electron Configuration
5.0 / 5 (0 votes)
