Worked example: Identifying an element from successive ionization energies | Khan Academy
Summary
TLDRIn this educational video, the instructor challenges viewers to identify a third period element based on its first five ionization energies. The script explains the concept of ionization energy and guides viewers through the process of elimination using a periodic table. The element in question, with three valence electrons and a significant increase in ionization energy for the fourth electron, is revealed to be aluminum.
Takeaways
- 🔍 The script discusses the ionization energies of a third period element's first five electrons.
- 📊 The first ionization energy is 578 kilojoules per mole, indicating the energy needed to remove the first electron from a neutral atom.
- 📈 The second ionization energy increases to 1,817 kilojoules per mole, and the third to 2,745 kilojoules per mole, showing a gradual increase.
- 🚀 The fourth ionization energy jumps significantly to 11,578 kilojoules per mole, suggesting a change in the type of electrons being removed.
- 🌟 The fifth ionization energy is even higher at 14,842 kilojoules per mole, further emphasizing the difficulty of removing inner electrons.
- 🔑 The increase in ionization energy for the first three electrons is expected as they are valence electrons.
- 🔬 The sharp increase after the third ionization suggests the removal of core electrons, which require more energy.
- 🧩 The element in question has three valence electrons, which is a key clue to its identity.
- 🌐 The periodic table is a valuable tool for identifying the element based on its ionization energies and electron configuration.
- 🎯 The element is identified as aluminum, which is in the third period and has three valence electrons.
- 📚 Understanding the periodic table and the concept of valence and core electrons is crucial for solving this problem.
Q & A
What is ionization energy?
-Ionization energy is the energy required to remove an electron from a neutral atom or ion to form a cation, and it is typically measured in kilojoules per mole (kJ/mol).
Why is it important to have a periodic table when trying to identify an element based on ionization energies?
-A periodic table is essential because it provides a visual representation of the elements' electron configurations, which can help in correlating the ionization energies with the number of valence electrons.
What does the first ionization energy of 578 kJ/mol suggest about the element?
-The first ionization energy of 578 kJ/mol suggests that the element has a relatively low energy barrier for losing its first electron, indicating it is likely in the earlier part of the third period of the periodic table.
How does the ionization energy trend for the first three electrons compare to the fourth and fifth?
-The ionization energy for the first three electrons increases gradually, but there is a significant jump for the fourth and fifth electrons, suggesting a change in the electron shell from which the electrons are being removed.
Why is there a large increase in ionization energy for the fourth electron compared to the third?
-The large increase in ionization energy for the fourth electron indicates that the electron is being removed from a more stable, fully filled second energy shell, which requires significantly more energy.
What is the significance of the term 'valence electrons' in the context of ionization energy?
-Valence electrons are the outermost electrons of an atom and are involved in chemical bonding. Their removal requires less energy compared to core electrons, which are more tightly bound to the nucleus.
Which third period element has three valence electrons?
-Aluminum (Al) is the third period element with three valence electrons, which aligns with the ionization energy pattern described in the script.
Why is aluminum the most likely element described in the script?
-Aluminum is the most likely element because its ionization energy pattern matches the script's description, with a gradual increase for the first three electrons and a significant increase for the fourth and fifth electrons.
What can we infer about the electron configuration of the mystery element from the ionization energy data?
-We can infer that the mystery element has three valence electrons and that the fourth and fifth ionization energies indicate the removal of electrons from a more stable, inner energy shell.
How does the concept of electron shells relate to ionization energy?
-Electron shells represent different energy levels within an atom. Electrons in higher energy shells (further from the nucleus) are easier to remove and thus require less ionization energy compared to those in lower energy shells.
What is the significance of the jump in ionization energy for the fourth and fifth electrons in identifying the element?
-The significant jump in ionization energy for the fourth and fifth electrons indicates that these electrons are being removed from a more stable, inner shell, which is a key clue in identifying the element as aluminum.
Outlines

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードMindmap

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードKeywords

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードHighlights

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードTranscripts

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレード関連動画をさらに表示

A Level Chemistry Revision "Ionisation Energy across a Period"
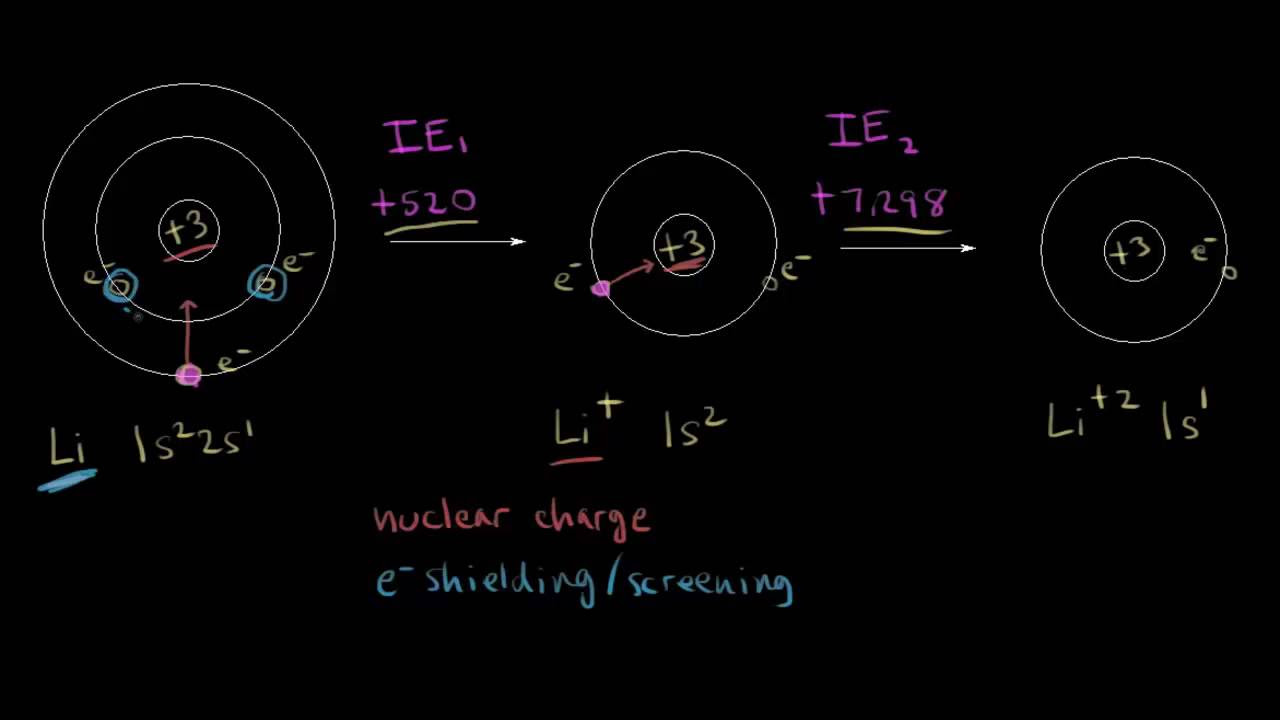
First and second ionization energy | Atomic structure and properties | AP Chemistry | Khan Academy
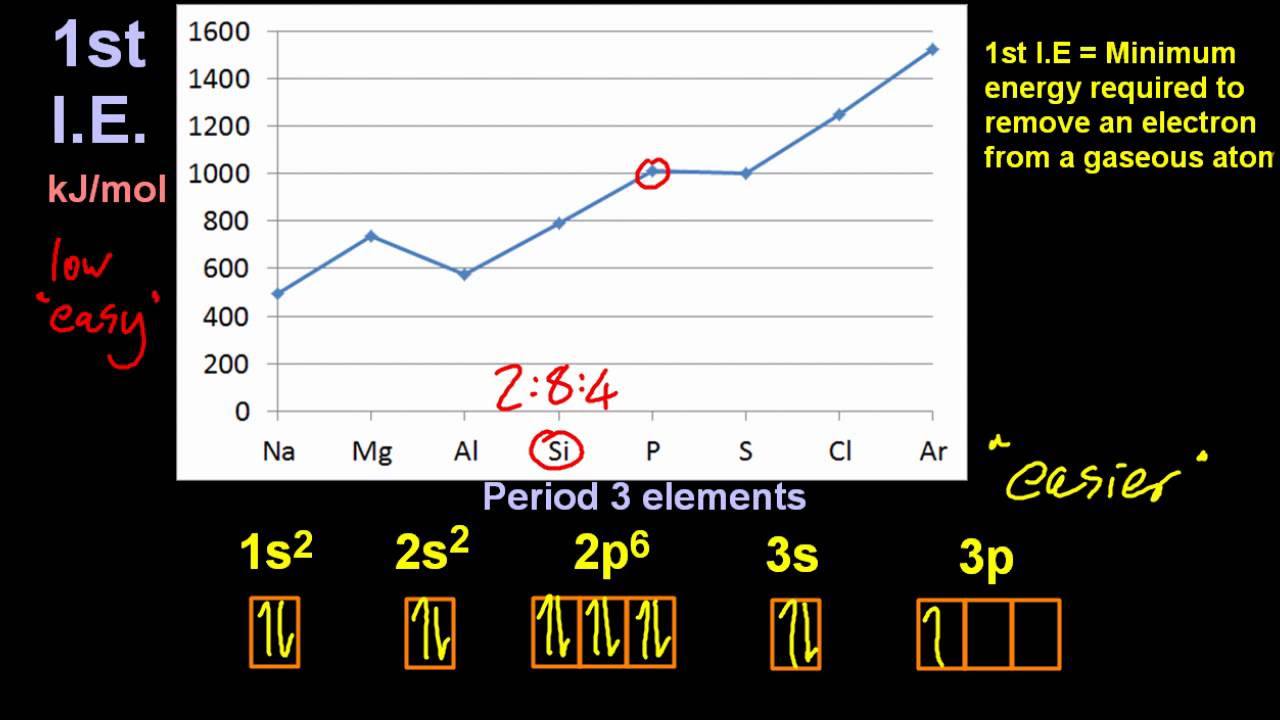
12.1/S1.3.6 Evidence of Energy Levels by 1st I.E. Trends Across a Period [HL IB Chemistry]

Introduction to photoelectron spectroscopy | AP Chemistry | Khan Academy

A Level Chemistry Revision "First Ionisation Energy"

Família e período da Tabela Periódica através da Distribuição Eletrônica.
5.0 / 5 (0 votes)
