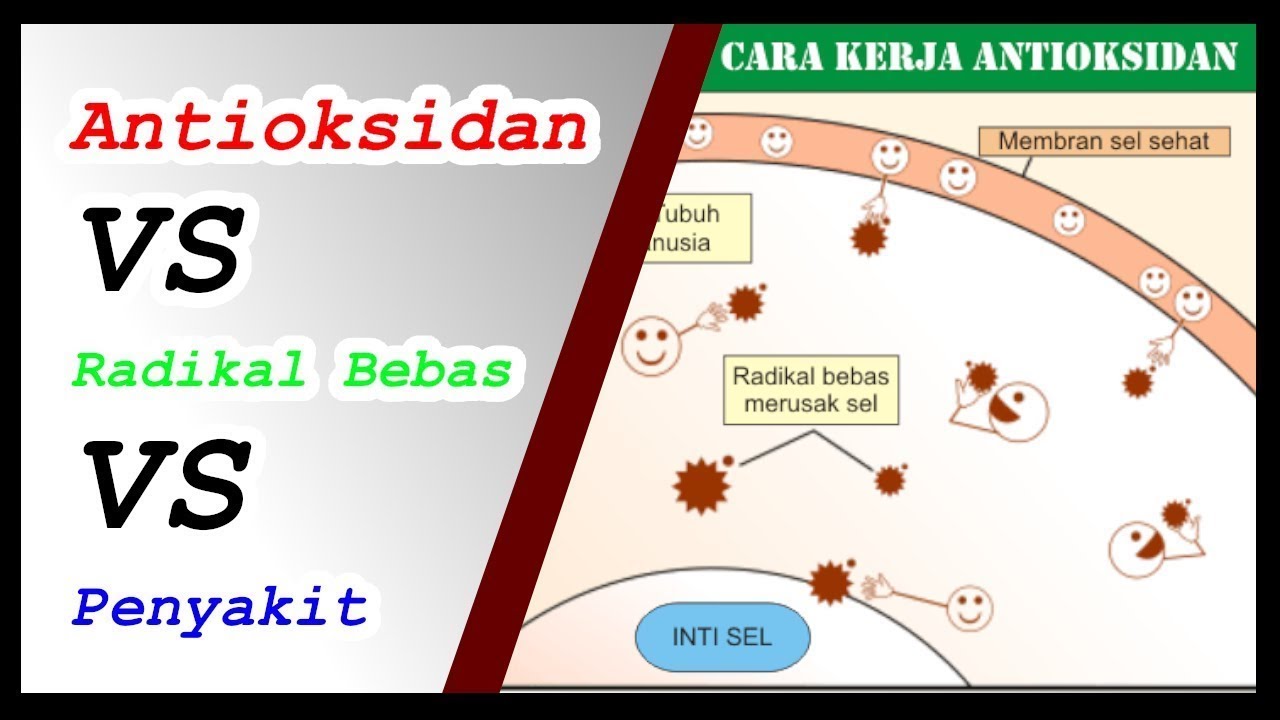
Antioxidants against Free Radicals [mechanisms]
Summary
TLDRIn this video, we explore the concept of free radicals—unstable, reactive molecules that can cause cellular damage and contribute to aging and various diseases. We also delve into how antioxidants protect the body by neutralizing these harmful radicals through various mechanisms, including hydrogen atom donation, electron donation, metal ion chelation, antioxidant enzyme regulation, and sirtuin activation. Examples such as resveratrol, vitamin C, and glutathione demonstrate how these antioxidants work in the body to maintain cellular health, reduce oxidative stress, and promote longevity.
Takeaways
- 😀 Free radicals are highly reactive molecules with unpaired electrons that can cause cellular damage, contributing to aging and diseases.
- 😀 Superoxide radicals, hydrogen peroxide, and hydroxyl radicals are some of the most common and harmful free radicals in the body.
- 😀 Oxidative stress occurs when there are excessive amounts of free radicals, leading to damage to lipids, proteins, and DNA.
- 😀 Antioxidants play a crucial role in protecting the body from free radical damage through various mechanisms.
- 😀 One of the main antioxidant mechanisms is hydrogen atom donation, where antioxidants like resveratrol stabilize free radicals by donating hydrogen atoms.
- 😀 Another mechanism is electron donation, where antioxidants like vitamin C donate electrons to free radicals, stabilizing them.
- 😀 Metal ion chelation is a process where antioxidants like glutathione bind to metal ions (e.g., iron and copper) to prevent harmful reactions like the Fenton reaction.
- 😀 Superoxide dismutase (SOD) is an important antioxidant enzyme that converts superoxide radicals into hydrogen peroxide and oxygen, reducing their harmful effects.
- 😀 Glutathione peroxidase breaks down hydrogen peroxide and lipid peroxides, helping to prevent their conversion into damaging radicals.
- 😀 Resveratrol activates sirtuins, which regulate antioxidant genes and DNA repair pathways, helping to protect cells from oxidative stress and support longevity.
Q & A
What are free radicals?
-Free radicals are highly reactive and unstable chemical species that contain one or more unpaired electrons in their outermost electron shell. These unpaired electrons make them seek to pair up with other electrons in nearby molecules, potentially causing damage to cells.
How do free radicals affect the body?
-Excessive amounts of free radicals can cause damage to cellular structures, including lipids, proteins, and DNA. This process, known as oxidative stress, is linked to various diseases and aging.
What is the superoxide radical, and how is it formed?
-The superoxide radical is a type of reactive oxygen species (ROS) formed when an oxygen molecule gains an extra electron. It is primarily produced in the mitochondria during oxidative phosphorylation when electrons leak from the electron transport chain and combine with molecular oxygen.
What are reactive oxygen species (ROS)?
-Reactive oxygen species (ROS) are chemically reactive molecules containing oxygen, such as superoxide radicals, hydrogen peroxide, and hydroxyl radicals. These species play roles in cellular metabolism but can be harmful in excess.
How does hydrogen peroxide form from superoxide radicals?
-Two superoxide radicals can react with two protons to produce one molecule of hydrogen peroxide and one molecule of oxygen. This reaction is catalyzed by the enzyme superoxide dismutase.
How do antioxidants help protect us from free radicals?
-Antioxidants protect the body from free radicals by neutralizing or stabilizing them through various mechanisms, such as hydrogen atom donation, electron donation, metal ion chelation, antioxidant enzyme regulation, and activation of sirtuins.
What is the role of resveratrol in antioxidant protection?
-Resveratrol, a polyphenolic compound, acts as an antioxidant by donating hydrogen atoms to free radicals, stabilizing them. This process transforms harmful radicals like the hydroxyl radical into stable, harmless molecules like water.
What is metal ion chelation, and how does it work?
-Metal ion chelation is the process by which certain antioxidants, such as glutathione, bind to metal ions like iron or copper. This prevents these metals from catalyzing reactions that would generate harmful hydroxyl radicals.
How does superoxide dismutase (SOD) function as an antioxidant enzyme?
-Superoxide dismutase (SOD) is an enzyme that catalyzes the conversion of superoxide radicals into hydrogen peroxide and oxygen. This reduces the reactivity of superoxide radicals, making them less harmful.
What is the role of glutathione peroxidase in combating oxidative stress?
-Glutathione peroxidase is an enzyme that helps break down hydrogen peroxide and lipid peroxides into harmless water and oxygen, thus preventing oxidative damage. It relies on glutathione, a tripeptide antioxidant, to function effectively.
How does resveratrol activate sirtuins, and why is this important?
-Resveratrol activates sirtuins, particularly Sirtuin 1, which promotes the expression of antioxidant genes and proteins, thereby enhancing the body's defense against oxidative stress. Sirtuin 1 also plays a role in DNA repair and cellular longevity.
Outlines

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードMindmap

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードKeywords

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードHighlights

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレードTranscripts

このセクションは有料ユーザー限定です。 アクセスするには、アップグレードをお願いします。
今すぐアップグレード5.0 / 5 (0 votes)