Mengenal GUGUS FUNGSI SENYAWA TURUNAN ALKANA, mulai dari Haloalkana sampai ESTER ‼️
Summary
TLDRThis chemistry lesson introduces the concept of functional groups in alkane derivatives, explaining how specific atoms or groups of atoms determine the properties of carbon compounds. It covers various functional groups such as haloalkanes, alcohols, ethers, aldehydes, ketones, carboxylic acids, and esters, detailing their structure and examples. The lesson also discusses different types of isomerism, including structural, positional, functional group, geometric, and optical isomerism, with practical applications in everyday life. The session wraps up with an invitation for further learning on alcohols and ethers in the next lesson.
Takeaways
- 🧪 Carbon compounds are extremely numerous, so chemists classify them based on shared characteristic properties.
- 🔬 Functional groups are specific atoms or groups of atoms that determine the characteristic properties and reactivity of carbon compounds.
- ⚛️ The functional group is the reactive part of an organic molecule and undergoes change during chemical reactions.
- 🧫 Haloalkanes are alkane derivatives in which a hydrogen atom is replaced by a halogen (F, Cl, Br, or I), with the general formula R–X.
- 🍷 Alcohols contain a hydroxyl (-OH) functional group attached to a carbon atom, with the general formula R–OH; ethanol is commonly used as an antiseptic.
- 💨 Ethers have the general structure R–O–R' and are commonly used as anesthetics.
- 🧴 Aldehydes (alkanals) contain the carbonyl group bonded to at least one hydrogen (R–CHO) and are used as disinfectants and preservatives.
- 🧯 Ketones (alkanones) contain a carbonyl group (C=O) bonded to two carbon atoms (R–CO–R'), such as propanone.
- 🍶 Carboxylic acids (alkanoic acids) contain the carboxyl group (-COOH) and are used in flavorings and fragrances; acetic acid is a common example.
- 🌸 Esters (alkyl alkanoates) contain the -COO- functional group and are widely used as solvents and in fragrances.
- 🧩 Naming alkane derivatives involves identifying the functional group, selecting the parent chain, numbering the chain, and determining possible isomers.
- 🔄 Structural isomerism in alkane derivatives includes chain isomerism, position isomerism, and functional group isomerism.
- 🔁 Stereoisomerism includes geometric (cis-trans) isomerism and optical isomerism.
- 🌍 Functional group chemistry has practical applications in daily life, such as ethanol for antiseptics and ethers for anesthesia.
Q & A
What are functional groups in organic chemistry?
-Functional groups are specific groups of atoms within molecules that determine the chemical reactivity and properties of the compound. They play a key role in classifying organic compounds.
Why are functional groups important in chemistry?
-Functional groups are important because they influence the chemical reactions of a compound. When an organic compound reacts with another substance, it is the functional group that undergoes changes.
What is Haloalkane, and how is it formed?
-Haloalkanes are organic compounds where a carbon atom is bonded with a halogen (like chlorine, bromine, fluorine, or iodine). They are formed when a halogen replaces a hydrogen atom in an alkane.
What is the general formula of Alcohol?
-The general formula of alcohol is R-OH, where 'R' represents a carbon chain, and the functional group is the hydroxyl group (-OH).
What is the difference between aldehyde and ketone?
-Aldehydes have a carbonyl group (-C=O) at the end of a carbon chain, whereas ketones have a carbonyl group in the middle of the carbon chain, bonded to two carbon atoms.
Give an example of an alcohol and its common use.
-An example of alcohol is ethanol (C2H5OH), which is commonly used as an antiseptic and in alcoholic beverages.
What is the significance of ester functional groups?
-Ester groups are formed from the reaction between alcohol and carboxylic acid. They are commonly used in fragrances, solvents, and in the food industry as flavoring agents.
What is isomerism in organic compounds?
-Isomerism is the occurrence of compounds with the same molecular formula but different structural arrangements. Isomers can differ in their physical and chemical properties.
What is the difference between cis-trans isomerism and optical isomerism?
-Cis-trans isomerism involves the arrangement of substituent groups around a double bond or ring structure, whereas optical isomerism refers to molecules that are non-superimposable mirror images of each other, often affecting their interaction with light.
Why are functional groups important in classifying organic compounds?
-Functional groups allow chemists to categorize organic compounds based on their reactivity and the type of bonds they form, making it easier to predict their chemical behavior in reactions.
Outlines

هذا القسم متوفر فقط للمشتركين. يرجى الترقية للوصول إلى هذه الميزة.
قم بالترقية الآنMindmap

هذا القسم متوفر فقط للمشتركين. يرجى الترقية للوصول إلى هذه الميزة.
قم بالترقية الآنKeywords

هذا القسم متوفر فقط للمشتركين. يرجى الترقية للوصول إلى هذه الميزة.
قم بالترقية الآنHighlights

هذا القسم متوفر فقط للمشتركين. يرجى الترقية للوصول إلى هذه الميزة.
قم بالترقية الآنTranscripts

هذا القسم متوفر فقط للمشتركين. يرجى الترقية للوصول إلى هذه الميزة.
قم بالترقية الآنتصفح المزيد من مقاطع الفيديو ذات الصلة

The Functional Group Concept Explained | Organic Chemistry | FuseSchool
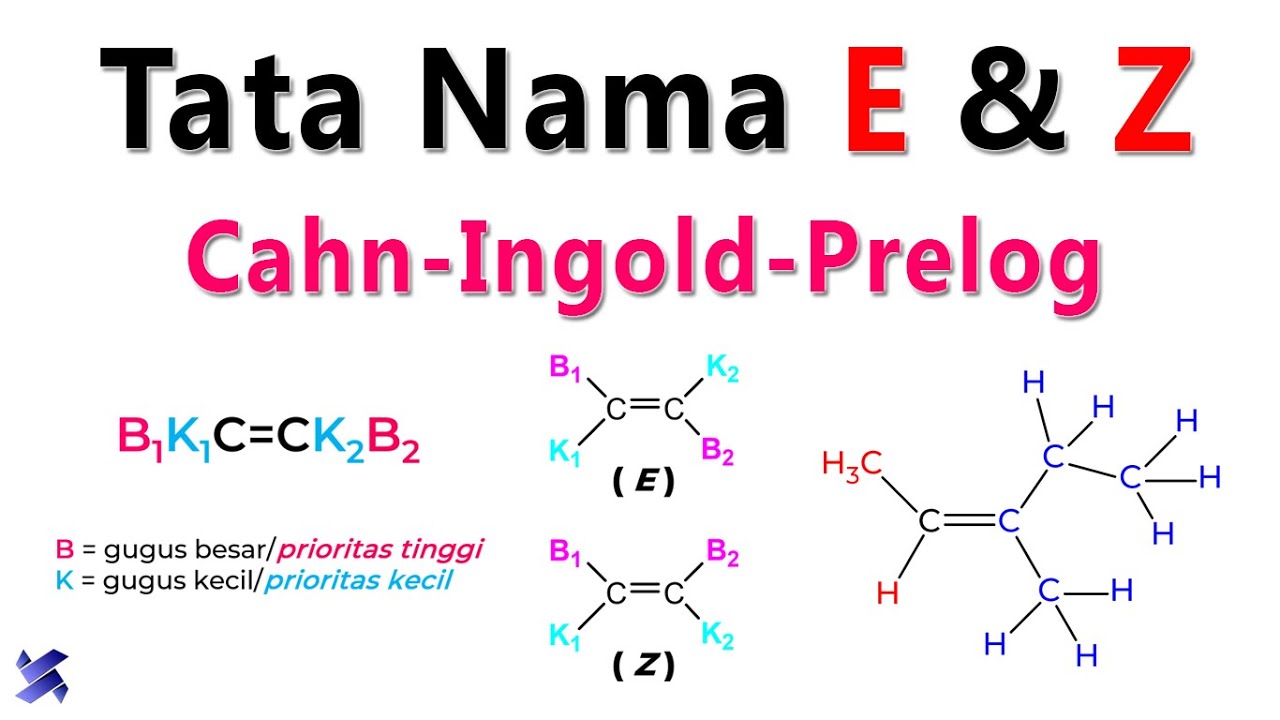
Isomer Geometri: Tata Nama E & Z dan Aturan Deret Prioritas Cahn-Ingold-Prelog

Nazewnictwo Alkany Węglowodory

Tata Nama Senyawa Turunan Alkana | KIMIA KELAS 12

Clase U12-Fundamentos de Química Orgánica-1° Parte-Alcanos

Introdução à química orgânica [Mapa Mental] Resumo
5.0 / 5 (0 votes)
