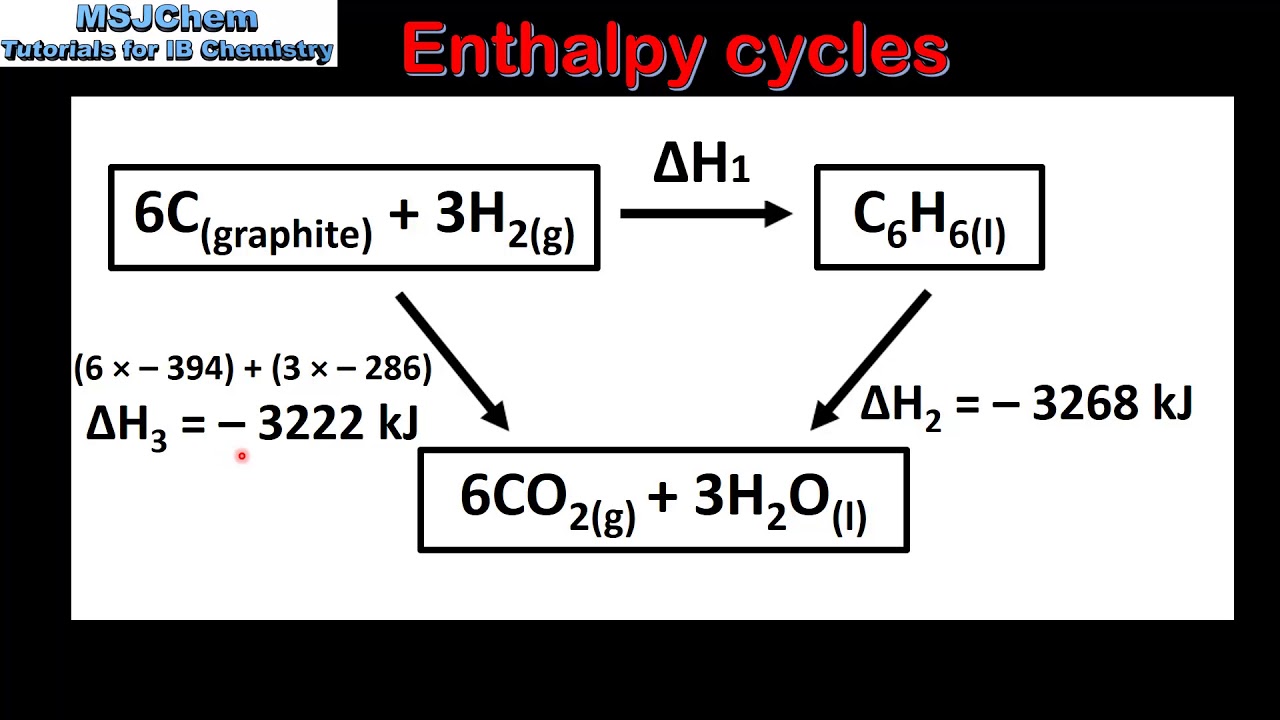
Practice Problem: Hess's Law
Summary
TLDRThis educational video script explains Hess's Law, a fundamental concept in thermodynamics, using a step-by-step approach to solve for the enthalpy change of a reaction. The script guides viewers through manipulating given thermochemical equations by adjusting stoichiometric coefficients and enthalpy values, ensuring that unwanted substances cancel out. The process involves halving coefficients and enthalpy changes, flipping reactions, and adding the modified enthalpy values to find the desired ΔH for the reaction ClF + F2 → ClF3. The final result is a clear demonstration of how to apply Hess's Law to determine the enthalpy change for a chemical reaction.
Takeaways
- 🔍 Hess's Law is used to calculate the change in enthalpy for a reaction when direct measurement is not possible.
- 📐 The given thermochemical data includes three reactions with known enthalpy changes.
- 🔄 To use Hess's Law, manipulate the given equations by reversing or adjusting coefficients to match the desired reaction.
- ✂️ Halving the coefficients in an equation also halves the corresponding enthalpy change (ΔH).
- 🔀 Reversing a reaction changes the sign of ΔH and adjusts the stoichiometric coefficients to match the desired direction.
- 🧩 Combine the manipulated equations to achieve the target reaction, ensuring that unwanted substances cancel out.
- 📉 When combining ΔH values, add them together based on their manipulated signs and magnitudes.
- 🌡️ The final ΔH for the target reaction is calculated by summing the adjusted ΔH values from the manipulated equations.
- ⚖️ Ensure that the reactants and products balance out correctly after combining the equations.
- 📝 The process involves careful manipulation of equations and ΔH values to isolate the desired reaction.
Q & A
What is Hess's Law and how is it used in the context of the given problem?
-Hess's Law states that the total enthalpy change for a chemical reaction is the same, regardless of the number of steps the reaction is carried out in. In the context of the problem, it is used to calculate the enthalpy change for a reaction where the direct enthalpy change is unknown by combining known enthalpy changes from other related reactions.
Why is it necessary to manipulate the stoichiometric coefficients and the ΔH values when applying Hess's Law?
-Manipulating the stoichiometric coefficients and ΔH values is necessary to align the reactants and products of the given reactions with the desired target reaction. This allows for the correct combination of the reactions to achieve the desired outcome, ensuring that unwanted substances cancel out and the target reaction is isolated.
How does the process of 'flipping' a reaction contribute to solving the problem?
-Flipping a reaction means reversing the direction of the reaction, which changes the products to reactants and vice versa. This is crucial when the desired species are present in the wrong state (i.e., as a reactant when needed as a product, or vice versa). By flipping, the reactions can be adjusted to match the required form for the target reaction.
What is the significance of cutting the stoichiometric coefficients and ΔH values in half in the given example?
-Cutting the stoichiometric coefficients and ΔH values in half is done to adjust the amount of a substance in a reaction to match the target reaction's requirement. In the example, ClF is needed in a 1:1 ratio, but the given reaction has it in a 2:1 ratio, so halving the reaction ensures the correct stoichiometry for the calculation.
Why is it important to ensure that unwanted substances cancel out when using Hess's Law?
-Ensuring that unwanted substances cancel out is important because it allows for the isolation of the desired reaction's enthalpy change. If substances do not cancel out, they would incorrectly contribute to the overall enthalpy change of the reaction, leading to an incorrect result.
How does the process of adding up the ΔH values from the manipulated reactions yield the enthalpy change for the target reaction?
-By correctly manipulating and aligning the given reactions to match the target reaction, the ΔH values from each step can be added together. Since enthalpy changes are state functions, the sum of the enthalpy changes from the individual steps will equal the enthalpy change for the overall reaction.
What is the final ΔH value calculated for the reaction ClF + F2 → ClF3 in the given example?
-The final ΔH value calculated for the reaction ClF + F2 → ClF3 is -139.2 kilojoules.
Why is it necessary to flip the sign of the ΔH value when flipping a reaction?
-Flipping the sign of the ΔH value is necessary because the enthalpy change is a measure of the energy absorbed or released by the system. When a reaction is reversed, the energy change associated with it is also reversed, thus the sign of ΔH must be flipped to reflect this change.
Can Hess's Law be used to determine the enthalpy change for any chemical reaction?
-Hess's Law can be used to determine the enthalpy change for any chemical reaction where the individual steps and their enthalpy changes are known. It is particularly useful when direct measurement of the enthalpy change for a reaction is not feasible.
What is the role of the tutorial mentioned in the script in understanding Hess's Law?
-The tutorial mentioned in the script serves as a supplementary resource for those unfamiliar with Hess's Law. It provides a detailed explanation of the concepts and techniques needed to solve problems like the one presented in the script.
Outlines

هذا القسم متوفر فقط للمشتركين. يرجى الترقية للوصول إلى هذه الميزة.
قم بالترقية الآنMindmap

هذا القسم متوفر فقط للمشتركين. يرجى الترقية للوصول إلى هذه الميزة.
قم بالترقية الآنKeywords

هذا القسم متوفر فقط للمشتركين. يرجى الترقية للوصول إلى هذه الميزة.
قم بالترقية الآنHighlights

هذا القسم متوفر فقط للمشتركين. يرجى الترقية للوصول إلى هذه الميزة.
قم بالترقية الآنTranscripts

هذا القسم متوفر فقط للمشتركين. يرجى الترقية للوصول إلى هذه الميزة.
قم بالترقية الآنتصفح المزيد من مقاطع الفيديو ذات الصلة
5.0 / 5 (0 votes)