Kuliah 1 Gravimetri
Summary
TLDRThis video lecture introduces gravimetric analysis, focusing on the process of determining calcium ion content in a solution. The method involves four stages: precipitation, filtration, drying, and weighing the precipitate. Calcium ions react with oxalate ions to form calcium oxalate, which is then filtered, heated, and weighed to calculate the mass of calcium. A stoichiometric calculation, involving the molar masses of calcium and calcium oxalate, determines the calcium concentration in the sample. The lecture also includes an example calculation, demonstrating how gravimetric methods are applied to analyze sample composition accurately.
Takeaways
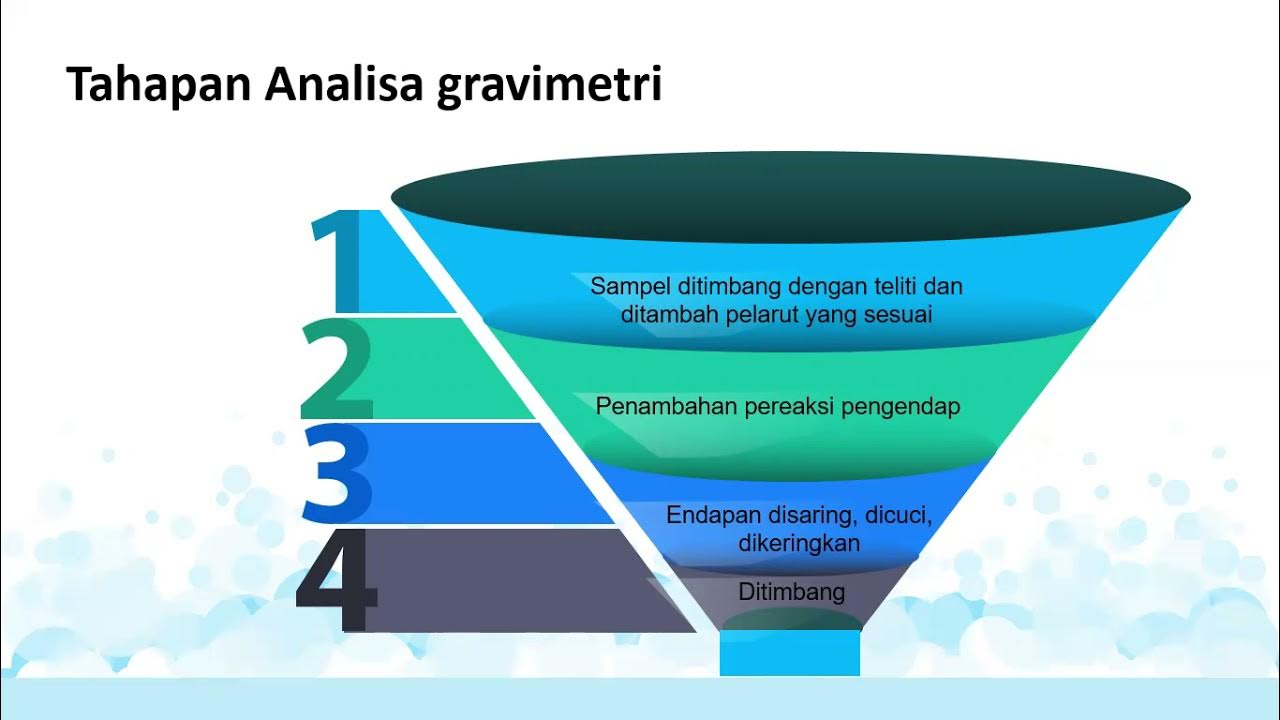
- 😀 Gravimetric analysis is a method based on measuring the mass of a substance to determine its quantity, with four key stages: precipitation, filtration, drying/ignition, and weighing.
- 😀 The process begins by adding a precipitating reagent (like oxalate ions) to a calcium-containing solution, which reacts to form calcium oxalate as a solid precipitate.
- 😀 The quality of the precipitate is important for accurate analysis. A pure, large crystal ensures better filtration and prevents analysis errors.
- 😀 Filtration involves removing the supernatant (the liquid above the precipitate) before filtering the solid to prevent clogging the filter paper.
- 😀 During filtration, filtered liquid (supernatant) is removed first, and the precipitate is washed with distilled water to ensure all solids are captured.
- 😀 After filtration, the calcium oxalate precipitate is placed in a porcelain crucible and heated using a Bunsen burner to remove moisture and impurities, leaving behind solid calcium oxide.
- 😀 The mass of the crucible with the dried precipitate is measured to calculate the mass of the calcium oxalate by subtracting the initial mass of the empty crucible.
- 😀 The amount of calcium in the sample is determined using stoichiometric calculations based on the mass of calcium oxalate obtained.
- 😀 The gravimetric factor (the ratio of the molar mass of calcium to calcium oxalate) is used to calculate the mass of calcium in the sample from the mass of calcium oxalate.
- 😀 In the example calculation, a sample weighing 0.2412 grams produces 0.1233 grams of calcium oxalate, leading to a calcium content of 15.91% in the sample.
Q & A
What is gravimetry?
-Gravimetry is an analytical method based on weighing or determining mass. It involves measuring the mass of an analyte (substance) by using precipitation and filtration techniques.
What are the four stages of gravimetric analysis?
-The four stages of gravimetric analysis are: 1) Precipitation, 2) Filtration of the precipitate, 3) Heating of the precipitate, and 4) Weighing of the precipitate.
How is calcium ion concentration determined in gravimetry?
-To determine the concentration of calcium ions, an oxalate ion solution is added to the sample. Calcium ions react with oxalate ions to form calcium oxalate precipitate. The mass of this precipitate is used to calculate the concentration of calcium.
Why must the calcium oxalate crystals be large in gravimetric analysis?
-Large crystals of calcium oxalate are preferred because they facilitate easier filtration and minimize errors in the analysis. The larger size helps ensure accurate separation during filtration.
How is filtration performed in gravimetric analysis?
-In filtration, a funnel containing filter paper is used to separate the precipitate from the liquid. Initially, the supernatant liquid is poured off before the precipitate is added. The precipitate is then rinsed with distilled water to ensure complete recovery.
What is the role of heating the precipitate in gravimetric analysis?
-Heating the precipitate in a porcelain crucible ensures that all residual moisture and any volatile compounds are removed, leaving behind a pure sample for accurate weighing.
What is the significance of measuring the weight of the crucible before and after heating?
-Measuring the weight of the crucible before and after heating allows the determination of the mass of the precipitate. The difference in weight is used to calculate the amount of calcium in the sample.
How do you calculate the mass of calcium in the sample using gravimetric analysis?
-The mass of calcium is determined using stoichiometric calculations. The mass of calcium oxalate precipitate is measured, and using the molar ratios between calcium and calcium oxalate, the mass of calcium is calculated.
What is a gravimetric factor, and why is it important?
-The gravimetric factor is the ratio of the molar mass of the analyte (in this case, calcium) to the molar mass of the precipitate (calcium oxalate). It is essential for converting the mass of the precipitate into the mass of the analyte (calcium).
How can the calcium content in a sample be calculated from the mass of calcium oxalate?
-To calculate the calcium content, the molar mass of calcium is divided by the molar mass of calcium oxalate, and this factor is multiplied by the mass of the calcium oxalate precipitate. This provides the mass of calcium in the sample.
Outlines

此内容仅限付费用户访问。 请升级后访问。
立即升级Mindmap

此内容仅限付费用户访问。 请升级后访问。
立即升级Keywords

此内容仅限付费用户访问。 请升级后访问。
立即升级Highlights

此内容仅限付费用户访问。 请升级后访问。
立即升级Transcripts

此内容仅限付费用户访问。 请升级后访问。
立即升级浏览更多相关视频

How to do Gravimetric Analysis in Chemistry (with calculations and examples!)

Ch 4 part 4 4 5 concentration of solutions

PEKI4205 Kimia Analitik I - Analisis Gravimetri

GRAVIMETRY ANALYSIS CALCULATION - PART 2
Analisa Gravimetri

Melaksanakan Analisis Gravimetri Konvensional Mengikuti Prosedur - Teknik Kimia
5.0 / 5 (0 votes)
