PHET SIMULATIONS || Isotopes and Atomic Mass - Duri Ismul Izzah
Summary
TLDRThis educational video introduces viewers to virtual labs using the Path app, focusing on experiments related to isotopes and atomic mass. The video explains fundamental concepts about atoms and isotopes, demonstrating how to explore various isotopes of elements like hydrogen, helium, lithium, and others. The process of adding and removing neutrons to determine different isotopes is showcased through interactive examples. The video is designed to help students grasp the concept of atomic structure and isotope variation through practical virtual experimentation.
Takeaways
- 😀 The video demonstrates how to use the Path application for virtual chemistry labs, accessible both online and offline.
- 😀 The application interface allows language selection, including Indonesian, and features multiple virtual lab simulations.
- 😀 The focus of the lesson is on learning about isotopes and atomic mass through virtual experiments.
- 😀 An atom consists of protons and neutrons in the nucleus, with electrons orbiting around it.
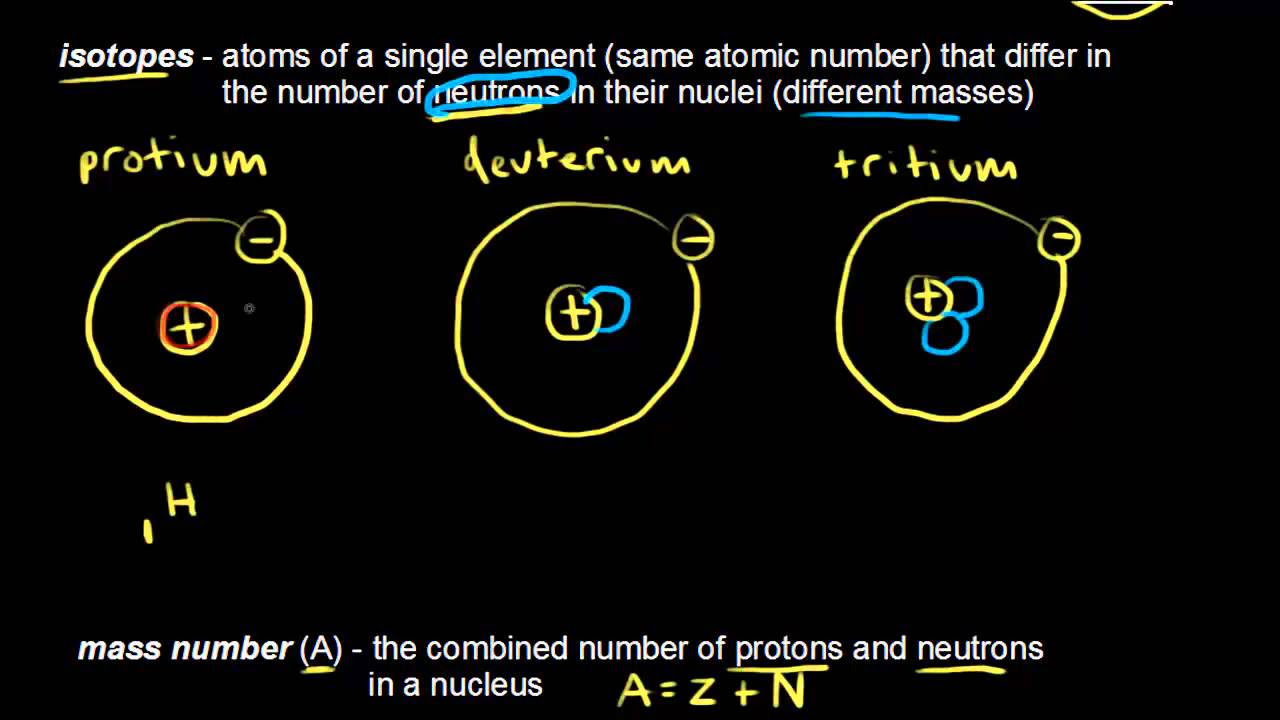
- 😀 Isotopes are atoms of the same element with the same number of protons but different numbers of neutrons, resulting in different atomic masses.
- 😀 The video shows how to modify neutron numbers in hydrogen, helium, lithium, beryllium, boron, carbon, nitrogen, oxygen, fluorine, and neon to observe their isotopes.
- 😀 Hydrogen isotopes include masses 1, 2, and 3; helium isotopes include masses 3 and 4.
- 😀 Carbon isotopes observed are 12, 13, and 14; oxygen isotopes are 16, 17, and 18.
- 😀 Fluorine has isotopes with masses 18 and 19; neon has isotopes with masses 20, 21, and 22.
- 😀 The video encourages viewers to remember atomic structure concepts before conducting virtual experiments and emphasizes interactive learning through the Path application.
Q & A
What is the main purpose of the video presented by Duri Izz?
-The main purpose of the video is to demonstrate how to use the virtual lab application 'PhET' to study isotopes and atomic mass of elements through simulations.
What are the two ways to access the PhET application mentioned in the video?
-PhET can be accessed in two ways: offline, by downloading the application, and online, directly through a web browser using Google.
How can the language of the PhET application be changed?
-The language in the PhET application can be changed to Indonesian by adjusting the language settings within the app.
What are the components of an atom according to the video?
-An atom consists of protons and neutrons located in the nucleus, and electrons that orbit around the nucleus.
How are isotopes defined in the video?
-Isotopes are defined as atoms of the same element that have the same number of protons but different numbers of neutrons, resulting in different atomic masses.
Which isotopes of hydrogen are identified in the simulation?
-The simulation identifies three isotopes of hydrogen: hydrogen-1, hydrogen-2 (deuterium), and hydrogen-3 (tritium), depending on the number of neutrons added.
What method is used in the video to determine the isotopes of an element?
-The method involves adding or removing neutrons in the PhET simulation to observe changes in atomic mass, which helps identify the isotopes.
Can you list the isotopes of carbon demonstrated in the video?
-The isotopes of carbon shown are carbon-12, carbon-13, and carbon-14, corresponding to the addition of neutrons in the simulation.
Why does the simulation sometimes not detect atomic mass when neutrons are added or removed?
-The simulation does not detect atomic mass when an impossible or unstable combination of neutrons and protons is attempted, reflecting that such isotopes do not naturally exist or are extremely unstable.
Which elements and their isotopes are explored in the video?
-The video explores the isotopes of hydrogen, helium, lithium, beryllium, boron, carbon, nitrogen, oxygen, fluorine, and neon, showing the changes in atomic mass when neutrons are added or removed.
What is the general observation about isotopes across different elements in the simulation?
-The general observation is that each element can have multiple isotopes with the same number of protons but varying numbers of neutrons, and adding or removing neutrons beyond certain limits results in undetectable atomic masses.
Outlines

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowMindmap

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowKeywords

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowHighlights

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowTranscripts

This section is available to paid users only. Please upgrade to access this part.
Upgrade NowBrowse More Related Video

What are Isotopes?
Atomic number, mass number, and isotopes | Chemistry | Khan Academy

MASSA ATOM RELATIF (Ar) DAN MASSA MOLEKUL RELATIF (Mr)

Struktur Atom (4) | Apa itu isotop, isoton, dan isobar? | Kimia Kelas 10

MASSA ATOM RELATIF : KIMIA SMA KELAS 10

Kimia SMA - Stokiometri (1) - Massa Atom Relatif, Massa Molekul Relatif, Ar, Mr (D)
5.0 / 5 (0 votes)