Atomic Structure and Bonding Explained | Chapter 2 – Materials Science and Engineering
Summary
TLDRThis video script explores the world of atomic structure and bonding, breaking down complex concepts into accessible insights. From the fascinating ability of geckos to walk on walls, thanks to atomic forces, to how diamonds form strong networks of atoms, the script connects these microscopic principles to real-world applications. It delves into the basics of atomic particles, quantum mechanics, and how electrons interact to form different types of bonds—ionic, covalent, and metallic. The script also touches on secondary bonds, like hydrogen bonds and Van der Waals forces, and emphasizes how understanding atomic structure can revolutionize material science, shaping everything from medical innovations to new materials.
Takeaways
- 😀 Geckos can walk on walls and ceilings thanks to millions of microscopic hairs on their feet, utilizing Van der Waals forces for grip.
- 😀 Understanding atomic structure and bonding can lead to breakthroughs in medicine, like bio-inspired adhesive tapes that could replace stitches in surgeries.
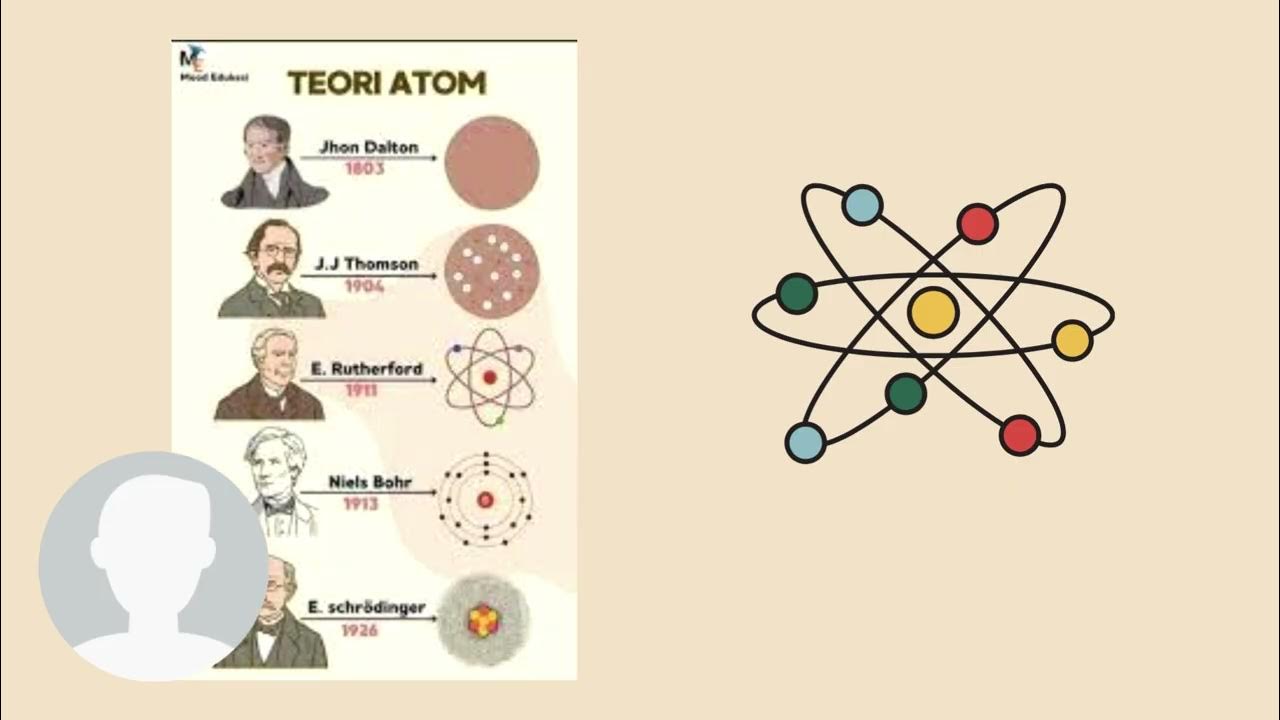
- 😀 Atoms consist of protons, neutrons, and electrons, with protons determining the element's identity and neutrons influencing its mass.
- 😀 Atomic mass comes from the combined mass of protons and neutrons, and isotopes are variations of elements with different neutron counts.
- 😀 Electrons are arranged in energy levels or orbitals, and their behavior is governed by quantum mechanics, where energy is quantized.
- 😀 Quantum numbers provide each electron with a unique ID, influencing how atoms interact and form bonds.
- 😀 The periodic table organizes elements based on electron arrangements, with elements in the same group sharing similar chemical properties.
- 😀 The three main types of chemical bonds are ionic, covalent, and metallic, each forming through different interactions of electrons.
- 😀 Ionic bonds form when electrons are transferred from one atom to another, creating positive and negative ions that attract each other.
- 😀 Covalent bonds involve the sharing of electrons, as seen in diamonds, where each carbon atom shares electrons with four neighbors to create a strong network.
Q & A
What is the main concept explored in the script?
-The main concept explored in the script is atomic structure and bonding, with a focus on how atomic interactions affect the properties of materials and have real-world applications, such as in gecko feet, diamonds, and medical innovations.
How do geckos manage to walk on walls and ceilings?
-Geckos can walk on walls and ceilings due to millions of microscopic hairs on their toes that use van der Waals forces to create weak attractions with surfaces. Although these forces are tiny individually, their cumulative effect is strong enough to support the gecko's entire weight.
What are van der Waals forces, and how are they important in nature?
-Van der Waals forces are weak attractions that occur between molecules or atoms due to temporary shifts in electron distribution, creating temporary dipoles. These forces are crucial in biological systems and materials science, such as how geckos can climb or how molecules interact in biological systems.
What is the difference between atomic number and atomic mass?
-The atomic number refers to the number of protons in an atom, which determines the element. Atomic mass, on the other hand, is the weighted average mass of an element's isotopes, taking into account the number of neutrons in each isotope.
What is the significance of quantum mechanics in atomic structure?
-Quantum mechanics is significant because it explains how electrons are arranged in specific energy levels or orbitals around the nucleus. Electrons can't exist between these energy levels, and their behavior is governed by quantum numbers, which determine how atoms will bond with each other.
How does the periodic table organize elements?
-The periodic table organizes elements based on their atomic number and the arrangement of electrons in their outermost shell. Elements in the same column share similar chemical properties due to their similar electron arrangements, which is why elements in a group behave similarly.
What makes ionic bonds different from covalent and metallic bonds?
-Ionic bonds involve the transfer of electrons from one atom to another, creating positive and negative ions that attract each other. Covalent bonds involve the sharing of electrons, while metallic bonds involve a 'sea' of free electrons shared among metal ions, giving metals properties like conductivity and malleability.
What are hybrid orbitals, and why are they important in molecule shape?
-Hybrid orbitals are formed when atomic orbitals combine to create new orbitals with different shapes and energy levels. These hybrid orbitals are essential for determining the shape and bond angles in molecules, influencing their properties and interactions with other molecules.
How do hydrogen bonds work, and why are they important in water?
-Hydrogen bonds occur when a hydrogen atom bonded to a highly electronegative atom (like oxygen) is attracted to another electronegative atom in a nearby molecule. In water, hydrogen bonds are crucial because they explain water's high boiling point, surface tension, and why ice floats on water.
What role do secondary bonds like van der Waals and hydrogen bonds play in materials and biology?
-Secondary bonds, like van der Waals forces and hydrogen bonds, are weaker than primary bonds but still significantly affect the properties of materials. They influence things like gas condensation, molecular interactions in biology, and the behavior of substances like water.
How does understanding atomic bonding impact material science and innovation?
-Understanding atomic bonding is the foundation of material science. By manipulating atomic structure and bonding, scientists can design new materials with specific properties, such as stronger alloys, flexible polymers, or heat-resistant ceramics, which can be used in industries like aerospace, medicine, and manufacturing.
Outlines

هذا القسم متوفر فقط للمشتركين. يرجى الترقية للوصول إلى هذه الميزة.
قم بالترقية الآنMindmap

هذا القسم متوفر فقط للمشتركين. يرجى الترقية للوصول إلى هذه الميزة.
قم بالترقية الآنKeywords

هذا القسم متوفر فقط للمشتركين. يرجى الترقية للوصول إلى هذه الميزة.
قم بالترقية الآنHighlights

هذا القسم متوفر فقط للمشتركين. يرجى الترقية للوصول إلى هذه الميزة.
قم بالترقية الآنTranscripts

هذا القسم متوفر فقط للمشتركين. يرجى الترقية للوصول إلى هذه الميزة.
قم بالترقية الآنتصفح المزيد من مقاطع الفيديو ذات الصلة
5.0 / 5 (0 votes)